Monitored Natural Attenuation - Transitioning from Active Remedies
Many contaminated sites use active remedies such as pump-and-treat or in situ remediation to clean up impacted groundwater. Natural attenuation processes such as natural degradation or hydrodynamic dispersion also contribute to the cleanup. As remediation progresses, a point is often reached when the time required to reach the remedial objectives using the active remedy is roughly the same as the time required if the active remedy is shut down, and the continuing remediation of the site is provided by natural attenuation processes alone. From that point forward, the extra effort and expense of the active remedy provides no benefit over natural attenuation, and it may be appropriate to transition the site to Monitored Natural Attenuation (MNA). This article deals with currently available tools and approaches that can be used to support a decision to transition from active remediation to MNA.
Contents
- 1 Introduction
- 2 Background on Rate Constants
- 3 Goals for MNA at Sites
- 4 When the Goal Applies to Point-of-Compliance Wells
- 5 When the Goal Applies to All the Wells
- 6 Extent of Treatment Necessary to Transition to MNA
- 7 The Transition Assessment Teaching Assistant (TA2) Tool
- 8 Summary
- 9 References
- 10 See Also
Related Article(s):
- Monitored Natural Attenuation (MNA)
- Alternative Endpoints
- Source Zone Modeling
- Plume Response Modeling
- REMChlor - MD
Contributor(s):
Key Resource(s):
Introduction
Many active remedies are effective at treating higher concentrations of contaminants, but as the contaminant concentrations decrease, the rate of cleanup may slow before the site reaches the cleanup goal. At some sites, the rate of cleanup may slow until it is not significantly different from the rate of cleanup provided by the natural attenuation processes that occur at the site. At other sites, the concentration of contaminants in water produced by a pumping system is below the cleanup goal, but the concentration in monitoring wells in the source area are still above the goal. At some sites, active treatment has stopped further expansion of the plume toward a receptor, and concentrations are declining over time throughout the plume, but back diffusion is sustaining concentrations in the plume that are above the cleanup goal.
In 2013, a significant National Research Council (NRC) report noted that despite years of effort and considerable investment, many sites “will require long-term management that could extend for decades or longer”[6]. The authors of the report discussed the need for developments that can aid in “transition from active remediation to more passive strategies and provide more cost-effective and protective long-term management of complex sites”[6].
The United States Environmental Protection Agency[7] allows the use of monitored natural attenuation (MNA) to attain the cleanup goals when the site-specific remediation objectives can be attained within a time frame that is reasonable compared to that offered by other more active methods. Many CERCLA[8] and RCRA[9] sites take advantage of this policy. An active remedy is typically used initially to treat high concentrations of contaminants followed by MNA to treat the lower concentrations that remain.
Unfortunately, there is no well-established approach to determine when it is appropriate to discontinue the active remedy. The NRC report[6] emphasized the use of more rigorous evaluations of existing data to support these efforts. This can include a quantitative assessment of the performance of active remedies (e.g., evidence of asymptotic performance) as well as documenting site conditions that may be contributing to these performance limitations. Importantly, it also identifies alternative approaches for managing the site, which could include MNA if the natural attenuation processes can meaningfully contribute to the achievement of site cleanup objectives.
This article reviews available tools and approaches to evaluate a transition to MNA. The tools and approaches depend on calculations of rate constants for natural attenuation with distance in flowing groundwater or rate constants for attenuation over time in individual monitoring wells.
Background on Rate Constants
At sites where a transition to MNA is being considered, a key step is estimating attenuation rate constants and understanding how they are extracted from monitoring data. A general formula to describe the rate of a chemical reaction is:
Equation 1: r = k [C] m where: r is the rate of the reaction, k is the rate constant, C is the concentration of the chemical undergoing the reaction, and the exponent m is the order of the reaction.
When the rate of the reaction is proportional to the concentration of the contaminant, the value of m is 1. Therefore, the reaction is described as a first-order reaction, and the rate constant is described as a first-order rate constant. In Equation 1, concentration could go up or down, but k is a constant of proportionality for the rate of increase in concentration. The rate constant for attenuation is the negative of k. If the rate of degradation is a fixed value regardless of concentration, the value of m is 0, and degradation is a zero-order process.
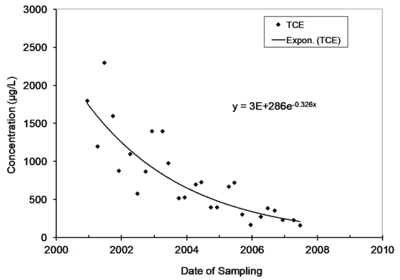
Natural attenuation of concentrations over time in monitoring wells is frequently described by a first-order rate constant, and natural biological or abiotic degradation of contaminants in flowing groundwater is typically also described by a first-order rate constant. Figure 1 provides an example of monitoring data that is described by a first-order rate constant.
The rate constant for attenuation over time in a single well and the rate constant for attenuation with distance along a flow path in an aquifer describe different situations that are controlled by different processes. Attenuation over time in a well is largely controlled by the rate of attenuation of the source of contamination in the aquifer. Attenuation with distance along a flow path includes attenuation of concentrations in the source along with contributions from biological degradation processes, abiotic degradation processes and hydrodynamic dispersion of the contaminated groundwater into clean groundwater[1].
The first-order rate constant for attenuation over time in a single well is commonly referred to as kpoint[1]. A time series chart in Microsoft EXCEL of the concentrations of a contaminant (y axis) on the date of sampling (x axis) can be used to extract a value for kpoint. Select the data, then insert an exponential trend line and display the equation on the chart. The value of kpoint can also be calculated in EXCEL using the Regression Analysis Tool in the Data Analysis Toolpak. Note that the rate constants extracted in EXCEL are constants for the rate of change, not the rate of attenuation. Take the negative of the rate of change to get kpoint. In the example in Figure 1, the unit of time on the X axis is years, and the value of kpoint is 0.326 per year.
Attenuation versus distance rate coefficients describe a bulk attenuation rate including both degradation and non-destructive processes such as dispersion. To extract values for rate constants for degradation alone, it is necessary to calibrate a groundwater flow and transport model to the data at the site. The model is calibrated with values for the hydrogeological properties of the aquifer (effective porosity, hydraulic gradient, hydraulic conductivity, hydrodynamic dispersion and the organic carbon content of the aquifer matrix). After the hydrogeological properties of the aquifer are fixed in the model, the most appropriate values for the degradation rate constants are the values that produce the best fit between the contaminant concentrations that are predicted by the model and the contaminant monitoring data at the site.
There are a number of reasons why natural attenuation processes are better described as first-order relationship instead of zero-order or some other order. The attenuation over time in a monitoring well tracks the attenuation over time of the source of contamination that sustains the plume[1]. Sites go through a lifecycle, and attenuation of sources at mature sites is often a first-order process[10]. If a chlorinated solvent site is mature, the contamination in the source area that was originally present as nonaqueous phase liquids (NAPL) has been redistributed and is now sequestered in a sorbed phase to aquifer solids or has diffused into non-transmissive portions of the aquifer matrix. Transfer of contaminants back into the more transmissive portions of the aquifer occurs by diffusion along a fixed path length, and the rate of transfer is controlled by the concentration of the contaminant remaining in the source material. Because the rate of transfer is proportional to the concentration of contaminant in the source material, attenuation of the source is a first-order process. These processes are discussed in more detail in Source Zone Modeling.
Degradation processes are also usually first order. Abiotic reactions are almost always first order with respect to the concentration of the target chemical. Biodegradation reactions are zero order at high concentrations because the available enzymes are saturated with substrate, but are first order at lower concentrations that are typical of natural attenuation conditions in groundwater.
Goals for MNA at Sites
The information necessary to evaluate whether a site can be transitioned to MNA depends on the goal for MNA at the site. For many cleanup actions, the goal is to confine contamination within a waste management area where the contamination is left in place, in which case the cleanup goal applies to point-of-compliance wells that are outside the waste management area. For other cleanup actions, the entire site must be cleaned up, in which case the cleanup goal applies to any monitoring well on the site. The time by which the goal is to be attained is specified at CERCLA sites in the Record of Decision (the ROD). At RCRA sites, the time allowed for the cleanup to be attained may be specified in the permit.
When the Goal Applies to Point-of-Compliance Wells
Consider the following framework for evaluating a transition to MNA:
- Use a computer model to extract rate constants for the natural degradation of the contaminant that occurred in groundwater at the site before the active remedy was installed.
- Assume that the same rate constants will apply after the active remedy is no longer in operation. Note that this assumption may not be valid if the active remedy changes the geochemistry of the aquifer in the flow path to the point-of-compliance well.
- Calibrate a computer groundwater flow and transport model with the hydrogeological properties of the aquifer that pertain after the active remedy is no longer in operation, the concentration of contaminant after the active remedy, and the rate constants for natural degradation that are assumed to apply after the active remedy.
- Use the computer model to project the concentrations of the contaminant at the point-of-compliance well over time.
- If the concentrations at the point-of-compliance wells are predicted to be less than the goal before the specified date, that is a quantitative line of evidence in support of a transition to MNA.
There are several computer applications that are particularly useful to extract rate constants at a site from monitoring data that were collected before the active remedy was installed. For example, Natural Attenuation Software (NAS)[2], BIOCHLOR[3] and BioPIC[4] can be downloaded from the internet at no cost. Another recent example, the TA2 Tool[5], is discussed in detail later in this article.
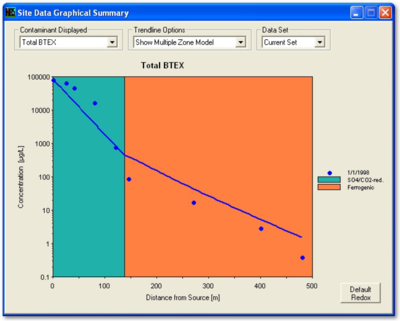
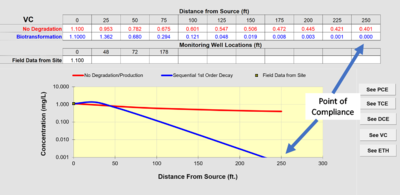
In NAS, the user inputs the hydrogeological data, the distance of wells along the flow path, and the concentrations of contaminants in the wells. The NAS application extracts rate constants and makes projections at the point-of-compliance. With NAS, it is possible to extract different rate constants for specific geochemical environments along the flow path.
Figure 2 provides an example calibration of NAS. The concentrations in the monitoring wells used to calibrate the model are compared to the simulation provided by the model. The values of the rate constants that are extracted from the field data are available in the “Output” tab under “Data and Results Table.”
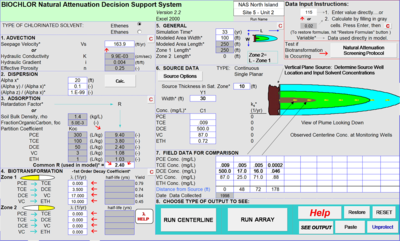
Figure 3 depicts the input screen for BIOCHLOR. The user inputs the hydrogeological parameters, the first-order rate constants (1st Order Decay Coefficient), the distribution of the wells along the flow path, and the concentrations of contaminants in the wells. The model is set up for conditions that apply before the installation of the active remedy.
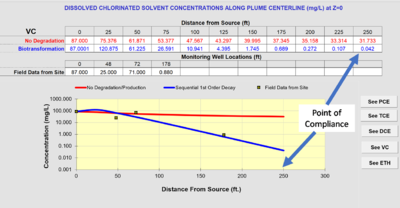
BIOCHLOR does not automatically fit the rate constants to the field data. Instead, the user examines the output of the model, and adjusts the rate constants until they provide the best fit between the model prediction and the monitoring data for wells at the site. This comparison is illustrated in Figure 4.
If the distance from the source well to the point-of-compliance well is set as the “Modeled Area Length” in Section 5 of the input screen, the “Run Centerline” output will provide the projected concentrations at that length. Assume the distance from the source well to the point-of-compliance well is 250 feet. The projected concentration in Figure 3 of vinyl chloride at a point-of-compliance well is 0.042 mg/L. If the regulatory goal were the federal drinking water maximum contaminant level (MCL)[11] of 0.002 mg/L, the projected concentration would exceed the goal, and MNA would not be adequate as a remedy.
For the sake of illustration, assume that an active remedy has been implemented, and the concentrations in the source well are 5.4 mg/L for DCE and 1.1 mg/L for vinyl chloride. To evaluate whether it is now appropriate to transition to MNA, BIOCHLOR could be calibrated with these concentrations to predict concentrations in the point-of-compliance well. (See Figure 5). In this example, the projected concentration at the point-of-compliance well does meet the goal.
Some active remedies are subject to rebound. If this is the case, the evaluation should begin at the point in time when it is clear that the trend in concentrations is downward.
A new EXCEL-based tool that does many of the same basic calculations as BIOCHLOR was recently developed as part of an update to the BioPIC[4] decision support software. This tool, the MNA Rate Constant Estimator, extracts rate constants from concentration versus distance data for a variety of different chemicals, including chlorinated ethenes (e.g., PCE and TCE), chlorinated ethanes (e.g., 1,1,1-TCA), and 1,4-dioxane. This tool was developed to run using current versions of EXCEL, whereas BIOCHLOR must be run using older versions of EXCEL that may be unavailable to many users. The MNA Rate Constant Estimator can be used to estimate degradation rate constants and/or predict plume footprints over time. Consequently, it is a useful addition to the BioPIC decision framework for understanding if MNA is appropriate remedy for a site, and it can also be helpful for estimating rate constants as part of a transition assessment.
When the Goal Applies to All the Wells
At sites where a concentration-based cleanup goal must be achieved at all wells, each well at the site is evaluated independently, and the rate constant that is applicable is the rate constant for attenuation over time in the well (kpoint). To evaluate whether the region in an aquifer that is sampled by a particular monitoring well is ready to transition to MNA, it is necessary to have monitoring data from a period of time before the remedy was implemented. This data is used to extract a value for kpoint in the aquifer under natural attenuation conditions. The evaluation of a transition to MNA will assume that the same value for kpoint will apply after the active remedy is complete. This assumption may not be appropriate if the active remedy caused a permanent change in the geochemistry of the aquifer. The assumption is usually appropriate for pump-and-treat remedies.
If kpoint before implementation of the active remedy describes the time course of natural attenuation after the active remedy is completed, the time required to attain the cleanup goal is predicted from the following:
ln ( Cgoal ) Equation 2: t = Ccurrent -kpoint where: Ccurrent is the current concentration after active remediation, Cgoal is the cleanup goal, and t is the time required for concentrations to attenuate from Ccurrent to Cgoal.
If the value of t estimated using Equation 2 is less than the difference between the current date and the date specified by the site stakeholders to attain the goal, that is evidence in support of a transition to MNA.
Some active remedies are subject to contaminant concentration rebound. If this is the case, the evaluation should use a value of Ccurrent that is attained after the rebound has stabilized.
This approach depends on a robust value for kpoint. It is worthwhile to do a sensitivity analysis on kpoint where the lower 95% or 90% confidence interval on kpoint is used in Equation 2 to see if that changes the outcome of the evaluation. The confidence intervals can be calculated in EXCEL using the Regression Analysis Tool in the Data Analysis Toolpak. Wilson[12] provides detailed discussion of the use of linear regression to extract kpoint and confidence intervals on kpoint. Wilson[12] also discusses the use of goodness-of-fit tests to determine if there is evidence that a first-order rate equation is not the best fit to the monitoring data, and as a result the use of Equation 2 would not be appropriate. The TA2 Tool[5] also has the capability to calculate kpoint with a user-specified confidence interval, as described below.
At many sites, there is no specified date when the cleanup goal must be attained. In this situation, the monitoring data can be evaluated to determine if the current rate of attenuation under the active remedy is faster than the rate of natural attenuation before the active remedy was installed. The monitoring data can be examined to identify a time interval when the benefit of the active remedy has approached an asymptote. A second value of for kpoint can be extracted for that time interval. The two values for kpoint can be evaluated statistically to see if the current rate is faster at some appropriate level of confidence. If there is no statistical evidence that the rate of attenuation is faster, that determination can support a decision to transition to MNA.
Extent of Treatment Necessary to Transition to MNA
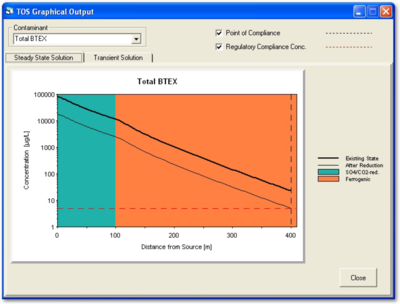
There are several computer applications that can predict the extent of treatment that must be achieved by the active remedy before it is worthwhile to evaluate the site for transition to MNA. For example, based on the distribution of contamination along the flow path, the NAS application will automatically predict a reduced concentration at the source well that will bring concentrations to the goal in the point-of-compliance well (Figure 6). A table that opens under the “DOS/TOS” tab provides the “Time of Equilibration” required to meet the goal at the reduced concentration. Modules in NAS allow the user to evaluate the effect of various pump-and-treat and source removal scenarios on the time required to attain the goal at the point-of-compliance well.
The REMChlor-MD[13] and REMFuel[14] models are flexible screening tools that allow a simultaneous evaluation of the extent of treatment provided by (1) source removal, (2) in situ remediation of the contaminated groundwater, or (3) natural attenuation processes in three discrete intervals along the flow path and three discrete time periods. Both REMChlor-MD[13] and REMFuel[14] can be downloaded from the internet at no cost. Liang et al.[15] provide a modeling program that uses Monte Carlo simulations to evaluate the effects of the uncertainties in the modeling parameters on the predictions of REMChlor-MD.
The Transition Assessment Teaching Assistant (TA2) Tool
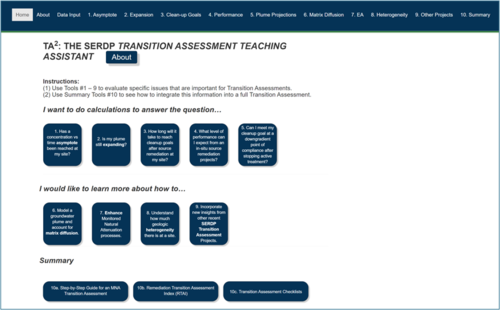
A learning and decision-making tool was recently released as part of Strategic Environmental Development and Research Program (SERDP) Project ER-201429 to help stakeholders gather information for the purposes of a site-specific transition assessment. This free software, the Transition Assessment Teaching Assistant (TA2) Tool[5], was developed using the elements identified in the 2013 NRC report[6] as the critical learning objectives for end users.
The Tool is a web-based app that includes a collection of individual modules designed to answer specific questions or research relevant topics (Figure 7). The Tool has been developed as an R Shiny app (version 1.8.0)[16], which is an interactive platform using R programming to perform all quantitative functions. The user can then view the results in a simple interface that easily accommodates plots, charts, and various mapping features in a Web browser. The Tool is free and does not require the user to install R software.
The modules within the TA2 Tool include:
Five Quantitative Tools that focus on assessing asymptotic groundwater concentrations from monitoring data, evaluating plume stability, estimating remediation timeframes after a hypothetical source removal project, forecasting remediation performance if a technology is applied in the field, or projecting concentrations at downgradient points of compliance.
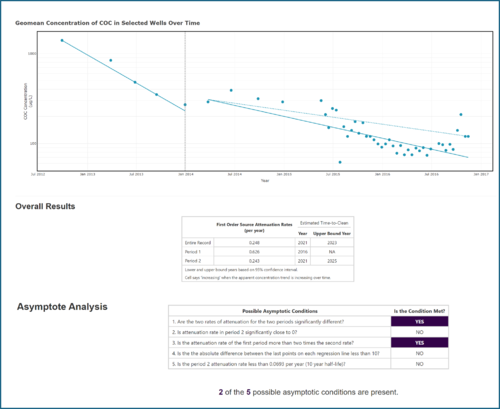
For example, Tool 1 in the TA2 Tool uses concentration versus time data from monitoring wells to estimate attenuation rate constants (kpoint) and evaluate if asymptotic conditions are present at particular locations or across the site. This helps to assess whether performance has plateaued at wells where a pump-and-treat system or other active treatment is in place. The user has the option to choose a “change point” within the monitoring record to determine if the attenuation rate has changed over time (e.g., once most of the accessible mass has been removed) (Figure 8). The user can either use visual interpretation to manually select the date when this apparent change occurred or have the date selected automatically using a binary segmentation protocol that is incorporated into the tool. The tool will calculate a rate for both the early period and a rate for the later period (after the change point), and then go through five different lines of evidence for asymptotic behavior (e.g., are the two rates of attenuation significantly different?). The user can then use the collective results as a technical justification demonstrating that the performance of the active remedy has plateaued as the first step in the transition assessment. The tool will also estimate the time to reach a user-specified cleanup goal if the overall attenuation rate (or the attenuation rate in the later period) were to continue.
Another module (Tool 5) focuses on evaluating sites where the concentration goal applies at a downgradient point of compliance, which is a key criterion for sites where MNA is being used as part of a risk-based strategy. The tool includes several different options to estimate a site-specific attenuation rate constant, including data from the pre-remediation period when natural attenuation processes were the sole means for reducing concentrations. Attenuation rate constants are then used to project the concentration versus distance from the contaminant source. Based on the predicted concentration at the downgradient point of compliance, the user can then see if the natural assimilative capacity along the aquifer flow path is sufficient to achieve the concentration goal in the absence of active treatment. For example, in the tab labeled “Use Pre-Remediation Rate Constant”, the logarithms of the concentrations from the period before active treatment began are plotted against the distance from the source well. The slope of the regression line is the rate constant for natural attenuation (including the contributions of degradation and dispersion). This rate constant can then be used to project the concentration moving downgradient from the well of concern after the end of active treatment. Similar approaches are provided within Tool 5 for using rate constants estimated from lab-based testing or derived from post-remediation data (after steady state has been reestablished).
Four Qualitative Tools provide information on matrix diffusion, enhanced attenuation options, geologic heterogeneity, and related research on transition assessments. Many of these modules are based on the current understanding of the role of matrix diffusion in influencing long-term concentration trends and remedial performance at contaminated groundwater sites. This includes summaries of different modeling options for better quantifying the effects of matrix diffusion. Sites impacted by matrix diffusion are generally challenging to treat using active remedies and thus are better candidates for less intensive management strategies that focus on reducing mass discharge rates, stabilizing the plume, and protecting potential downgradient receptors. As a result, matrix diffusion is critical to understanding and quantifying how natural attenuation processes are contributing to concentration trends.
One Summary Tool (Tool 10) compiles metrics from the other tools into a “Remediation Transition Assessment Index” (RTAI) and provides additional guidance on conducting site-specific transition assessments. The RTAI is a simple metric with a value from 1 to 5, where higher values reflect greater persistence of contamination due to matrix diffusion and other site-specific factors. An RTAI value is assigned to each of the results from the different tools that have been completed by the user. An RTAI of 5 suggests that the site is a strong candidate for transitioning to MNA or enhanced attenuation approaches, while a site with an RTAI value of 1 is a poor candidate. The user can assign an overall RTAI for the site based on the preponderance of evidence after reviewing the RTAI values generated by each tool, or calculate a site RTAI based on simple averaging, weighting, or other methods.
Tool 10 also contains a flowchart and a checklist for performing site-specific transition assessments that start with evaluating relevant bright line criteria, such as (1) can the concentration goals be met at the point of compliance by MNA; and (2) is the remediation timeframe for MNA reasonable and/or similar to the timeframe if source remediation were used. This checklist ensures that the user has gathered all relevant information that would be needed to support a technically rigorous site-specific Transition Assessment.
The TA2Tool provides a framework for remedial decision makers to evaluate different types of sites, including those where active treatment (e.g., pump and treat) is in use, as well as sites where future active source zone remediation is being considered. It also includes a description of enhanced MNA alternatives for sites where MNA alone may not be sufficient to control risk. As shown in Figure 8, the tool can be used to answer specific questions that have a primarily quantitative basis or to provide focused qualitative information for researching specific topics. Users can engage with just the modules that might be pertinent to assessment of an individual site, or they can go through all the modules to perform a more thorough, step-by-step analysis of the relevant issues for their site.
Summary
Tools and approaches are available that can be adapted to determine when a site is ready to transition from active remedy to MNA. However, these tools and approaches have not been applied for this purpose at a significant number of sites, and at the present time, they are not generally accepted by regulatory authorities. There is an opportunity to establish and implement a logical and consistent framework that can be widely implemented to evaluate sites for transition from active remedy to MNA.
References
- ^ 1.0 1.1 1.2 1.3 Newell, C.J., Rifai, H.S., Wilson, J.T., Connor, J.A., Aziz, J.A., Suarez, M.P., 2002. Calculation and Use of First-Order Rate Constants for Monitored Natural Attenuation Studies. 28p. EPA/540/S-02/500. Report.pdf
- ^ 2.0 2.1 Widdowson, M.A., Mendez, E., Chapelle, F.H., Casey, C.C., 2008. Natural Attenuation Software (NAS) Version 2.3.3. Virginia Polytechnic Institute and State University, the United States Geological Survey, and the United States Naval Facilities Engineering Command. NAS webpage: https://www.nas.cee.vt.edu/index.php See also: https://toxics.usgs.gov/highlights/nas_2.2.0/index.html
- ^ 3.0 3.1 Aziz, C.E., Newell, C.J. and Gonzales, J.R., 2002. BIOCHLOR Natural Attenuation Decision Support System Version 2.2 User’s Manual Addendum. Groundwater Services, Inc., Houston, Texas for the Air Force Center for Environmental Excellence.Report.pdf Available at: https://www.epa.gov/water-research/biochlor-natural-attenuation-decision-support-system
- ^ 4.0 4.1 4.2 Danko, A., Adamson, D., Newell, C., Wilson, J., Wilson, B., Freedman, D., Lebrón, C., 2021. Quick BioPIC User’s Guide, ESTCP Project ER-201730. Project Website User’s Guide
- ^ 5.0 5.1 5.2 5.3 Adamson, D.T., Newell, C.J., Hort, H.M, Wilson, J.T., 2024. TA2: The SERDP Transition Assessment Teaching Assistant. Strategic Environmental Research and Development Program (SERDP) Project ER20-1429. Project Website Online Tool
- ^ 6.0 6.1 6.2 6.3 National Research Council (NRC), 2013. Alternatives for Managing the Nation's Complex Contaminated Groundwater Sites. Committee on Future Options for Management in the Nation's Subsurface Remediation Effort, Water Science, Technology Board, Division on Earth and Life Studies, NRC. National Academies Press, 422 pages, ISBN 978-0-309-27874-4 doi: 10.17226/14668. Report.pdf
- ^ U.S. Environmental Protection Agency (USEPA), 1999. Use of Monitored Natural Attenuation at Superfund, RCRA Corrective Action, and Underground Storage Tank Sites. OSWER Directive 9200.4-17P. 39pp.Report.pdf
- ^ U.S. Environmental Protection Agency (USEPA), 2019. Summary of the Comprehensive Environmental Response, Compensation, and Liability Act (Superfund) https://www.epa.gov/laws-regulations/summary-comprehensive-environmental-response-compensation-and-liability-act
- ^ U.S. Environmental Protection Agency (USEPA), 2019. Resource Conservation and Recovery Act (RCRA) Laws and Regulations https://www.epa.gov/rcra
- ^ Sale, T., Newell, C., Stroo, H., Hinchee, R. and Johnson, P., 2008. Frequently Asked Questions Regarding Management of Chlorinated Solvents in Soils and Groundwater. Environmental Security Technology Certification Program (ESTCP, Project ER-200530), Department of Defense (DoD), Arlington, VA. Report.pdf Project Overview Website: https://www.serdp-estcp.org/Program-Areas/Environmental-Restoration/Contaminated-Groundwater/Persistent-Contamination/ER-200530
- ^ U. S. Environmental Protection Agency (USEPA), 2009. National Primary Drinking Water Regulations. EPA 816-F-09-004. Report.pdf
- ^ 12.0 12.1 Wilson, J.T. 2011. An Approach for Evaluating the Progress of Natural Attenuation in Groundwater. EPA 600-R-11-204. Report.pdf
- ^ 13.0 13.1 Falta, R.W., Farhat, S.K., Newell, C.J. and Lynch, K., 2018. A Practical Approach for Modeling Matrix Diffusion Effects in REMChlor. SERDP/ESTCP Project ER-201426 Report.pdf Website: https://www.serdp-estcp.org/Program-Areas/Environmental-Restoration/Contaminated-Groundwater/Persistent-Contamination/ER-201426
- ^ 14.0 14.1 Falta, R.W., Ahsanuzzaman, A.N., Stacy, M.B., Earle, R.C. and Wilson, J.T., 2012. Remediation Evaluation Model for Fuel Hydrocarbons (REMFuel). Users Manual Version 1.0. U.S. Environmental Protection Agency. EPA/600/R-12/028. Report.pdf Website: https://www.epa.gov/water-research/remediation-evaluation-model-fuel-hydrocarbons-remfuel
- ^ Liang, H., Falta, R.W., Newell, C.J., Farhat, S.K., Rao, P.S. and Basu, N., 2010. Decision & Management Tools for DNAPL Sites: Optimization of Chlorinated Solvent Source and Plume Remediation Considering Uncertainty. SERDP/ESTCP Project ER-200704. Report.pdf Project Overview Website: https://serdp-estcp.org/Program-Areas/Environmental-Restoration/Contaminated-Groundwater/Persistent-Contamination/ER-200704/(language)/eng-US
- ^ Chang, W., Cheng, J., Allaire, J., Sievert, C., Schloerke, B., Xie, Y., Allen, J., McPherson, J., Dipert, A., Borges, B., 2023. shiny: Web Application Framework for R. R package version 1.8.0, https://github.com/rstudio/shiny, https://shiny.posit.co/
See Also
- Newell, C.J., Kueper, B.H., Wilson, J.T., Johnson, P.C., 2014. Natural Attenuation of Chlorinated Solvent Source Zones. In: Chlorinated Solvent Source Zone Remediation, Editors: Kueper, B.H., Stroo, H.F., Vogel, C.M., Ward. SERDP ESTCP Environmental Remediation Technology, vol 7. Springer, New York, NY. pgs. 459-508. doi: 10.1007/978-1-4614-6922-3
- Kram, Mark, and Widdowson, Mark, 2008. Estimating Cleanup Times Associated with Combining Source-Area Remediation with Monitored Natural Attenuation. ESTCP ER-200436