Difference between revisions of "Metal and Metalloids - Remediation"
m (1 revision imported) |
m (1 revision imported) |
||
| Line 13: | Line 13: | ||
'''Key Resource(s)''': | '''Key Resource(s)''': | ||
| − | *[ | + | *[[Media:Evanko-1997-Remed_of_Metals.pdf|Remediation of Metals Contaminated Soils and Groundwater]]<ref name="Evanko1997">Evanko, C.R. and Dzombak, D.A., 1997. Remediation of metals-contaminated soils and groundwater. TE-97-01. Ground-water remediation technologies analysis center. [[Media:Evanko-1997-Remed_of_Metals.pdf|Report pdf]]</ref> |
*Groundwater and Soil Cleanup: Improving Management of Persistent Contaminants<ref name= "NAS1999">National Research Council, 1999. Groundwater and soil cleanup: Improving management of persistent contaminants. National Academies Press.</ref> | *Groundwater and Soil Cleanup: Improving Management of Persistent Contaminants<ref name= "NAS1999">National Research Council, 1999. Groundwater and soil cleanup: Improving management of persistent contaminants. National Academies Press.</ref> | ||
| − | *[ | + | *[[Media:TRUEX-2011-Scenarios_Approach_to_Attenuation-Based_Remedies.pdf|The Scenarios Approach to Attenuation-Based Remedies for Inorganic and Radionuclide Contaminants]]<ref name="Truex2011">Truex, M., Brady, P., Newell, C.J., Rysz, M., Denham, M., Vangelas, K. 2011. The scenarios approach to attenuation-based remedies for inorganic and radionuclide contaminants. Savannah-River National Laboratory U.S. Department of Energy. [[Media:TRUEX-2011-Scenarios_Approach_to_Attenuation-Based_Remedies.pdf|Report pdf]]</ref> |
==Introduction== | ==Introduction== | ||
| Line 37: | Line 37: | ||
!style="background-color:#CEE0F2;"|Technology (Target Media)!!style="background-color:#CEE0F2;"|Description !!style="background-color:#CEE0F2;"|Advantages!!style="background-color:#CEE0F2;"|Disadvantages !!style="background-color:#CEE0F2;"|Reference(s) | !style="background-color:#CEE0F2;"|Technology (Target Media)!!style="background-color:#CEE0F2;"|Description !!style="background-color:#CEE0F2;"|Advantages!!style="background-color:#CEE0F2;"|Disadvantages !!style="background-color:#CEE0F2;"|Reference(s) | ||
|- | |- | ||
| − | | Excavation (Contaminated soils)|| Removal of contaminated soils in unsaturated zone to eliminate secondary source of contamination.|| Widely available and accepted.|| Creates large amount of solid waste; not feasible in some geology; expense increases significantly for excavation below the water table.|| Post et al., 2013<ref>Post, T.C., Strom, D. and Beulow, L., 2013, July. The 100-C-7 Remediation Project. An Overview of One of DOE's Largest Remediation Projects-13260. WM Symposia, Tempe, AZ, 85282 (United States). [ | + | | Excavation (Contaminated soils)|| Removal of contaminated soils in unsaturated zone to eliminate secondary source of contamination.|| Widely available and accepted.|| Creates large amount of solid waste; not feasible in some geology; expense increases significantly for excavation below the water table.|| Post et al., 2013<ref>Post, T.C., Strom, D. and Beulow, L., 2013, July. The 100-C-7 Remediation Project. An Overview of One of DOE's Largest Remediation Projects-13260. WM Symposia, Tempe, AZ, 85282 (United States). [[Media:Post-2013-100-C-7_Remediation_Project.pdf|Report pdf]]</ref> |
|- | |- | ||
| − | | Pump-and-Treat (Groundwater plumes)|| Extraction of contaminated groundwater by well network for treatment at surface.|| Widely available and accepted.|| Surface treatment system can be expensive; can require long-time frames to reach remedial goals because of sorption of metals and metalloids.|| Mackay and Cherry, 1989<ref>Mackay, D.M. and Cherry, J.A., 1989. Groundwater contamination: Pump-and-treat remediation. Environmental Science & Technology, 23(6), 630-636. [http://dx.doi.org/10.1021/es00064a001 doi: 10.1021/es00064a001]</ref>; Mercer et al., 1990<ref>Mercer, J.W., Skipp, D.C. and Giffin, D., 1990. Basics of pump-and-treat ground-water remediation technology, pgs. 1-66. EPA-600/8-90/003. Robert S. Kerr Environmental Research Laboratory, Office of Research and Development, US Environmental Protection Agency. [ | + | | Pump-and-Treat (Groundwater plumes)|| Extraction of contaminated groundwater by well network for treatment at surface.|| Widely available and accepted.|| Surface treatment system can be expensive; can require long-time frames to reach remedial goals because of sorption of metals and metalloids.|| Mackay and Cherry, 1989<ref>Mackay, D.M. and Cherry, J.A., 1989. Groundwater contamination: Pump-and-treat remediation. Environmental Science & Technology, 23(6), 630-636. [http://dx.doi.org/10.1021/es00064a001 doi: 10.1021/es00064a001]</ref>; Mercer et al., 1990<ref>Mercer, J.W., Skipp, D.C. and Giffin, D., 1990. Basics of pump-and-treat ground-water remediation technology, pgs. 1-66. EPA-600/8-90/003. Robert S. Kerr Environmental Research Laboratory, Office of Research and Development, US Environmental Protection Agency. [[Media:Mercer-1990-Basics_of_Pump_%26_Treat.pdf|Report pdf]]</ref> |
|- | |- | ||
| Electrokinetics | | Electrokinetics | ||
(Contaminated fine grained material such as clays) | (Contaminated fine grained material such as clays) | ||
| − | ||Deployment of electrodes in subsurface to create an electrical field that drives contaminants to the electrodes.|| Effective in clay-rich aquifers; potential for less solid waste than excavation or pump-and-treat.|| Rarely used; increase of pH near cathode causing precipitation of metal salts; efficiency decreases outside of specific aquifer and contamination conditions.|| Van Cauwenberghe, 1997<ref>Van Cauwenberghe, L., 1997. Electrokinetics. TO-97-03. Ground-water remediation technologies analysis center. [ | + | ||Deployment of electrodes in subsurface to create an electrical field that drives contaminants to the electrodes.|| Effective in clay-rich aquifers; potential for less solid waste than excavation or pump-and-treat.|| Rarely used; increase of pH near cathode causing precipitation of metal salts; efficiency decreases outside of specific aquifer and contamination conditions.|| Van Cauwenberghe, 1997<ref>Van Cauwenberghe, L., 1997. Electrokinetics. TO-97-03. Ground-water remediation technologies analysis center. [[Media:Van_Cauwenberghe-1997-Electrokinetics.pdf|Report pdf]]</ref>; Virkutyte et al., 2002<ref>Virkutyte, J., Sillanpää, M. and Latostenmaa, P., 2002. Electrokinetic soil remediation—critical overview. Science of the Total Environment, 289(1), 97-121. [http://dx.doi.org/10.1016/s0048-9697(01)01027-0 doi: 10.1016/S0048-9697(01)01027-0]</ref>; Vocciante et al., 2016<ref>Vocciante, M., Caretta, A., Bua, L., Bagatin, R. and Ferro, S., 2016. Enhancements in ElectroKinetic Remediation Technology: Environmental assessment in comparison with other configurations and consolidated solutions. Chemical Engineering Journal, 289, 123-134. [http://dx.doi.org/10.1016/j.cej.2015.12.065 doi:10.1016/j.cej.2015.12.065]</ref> |
|- | |- | ||
| Phytoextraction | | Phytoextraction | ||
(Mostly contaminated soils, sometimes groundwater) | (Mostly contaminated soils, sometimes groundwater) | ||
| − | || Use of plants to extract contaminants from the subsurface.|| Only periodic maintenance (harvesting and processing of plants or plant detritus) once plants are established.|| Need long-term access to treat soils; difficult to treat deep groundwater; requires hyperaccumulating plants that may not exist for metals that are not essential nutrients.|| Pivetz, 2001<ref>Pivetz, B.E., 2001. Phytoremediation of Contaminated Soil and Ground Water at Hazardous Waste Sites. EPA/540/S-01/500. US Environmental Protection Agency. [ | + | || Use of plants to extract contaminants from the subsurface.|| Only periodic maintenance (harvesting and processing of plants or plant detritus) once plants are established.|| Need long-term access to treat soils; difficult to treat deep groundwater; requires hyperaccumulating plants that may not exist for metals that are not essential nutrients.|| Pivetz, 2001<ref>Pivetz, B.E., 2001. Phytoremediation of Contaminated Soil and Ground Water at Hazardous Waste Sites. EPA/540/S-01/500. US Environmental Protection Agency. [[Media:Pivetz-2001-Phytoremediation.pdf|Report pdf]]</ref>; McGrath and Zhao, 2003<ref>McGrath, S.P. and Zhao, F.J., 2003. Phytoextraction of metals and metalloids from contaminated soils. Current Opinion in Biotechnology, 14(3), 277-282. [http://dx.doi.org/10.1016/s0958-1669(03)00060-0 doi: 10.1016/S0958-1669(03)00060-0]</ref>; Sheoran et al., 2016<ref>Sheoran, V., Sheoran, A.S. and Poonia, P., 2016. Factors affecting phytoextraction: A review. Pedosphere, 26(2), 148-166. [http://dx.doi.org/10.1016/S1002-0160(15)60032-7 doi: 10.1016/S1002-0160(15)60032-7]</ref> |
|- | |- | ||
|} | |} | ||
| − | Improving the efficiency and reducing the cost of excavation, pump-and-treat, electrokinetics, and phytoextraction remains an important pursuit. Sometimes, the technologies in Table 1 can be combined to reduce costs. For excavation, the primary focus has been on reducing the volume of contaminated soil needing special disposal. This can involve soil washing<ref>ITRC, 1997. Technical and Regulatory Guidelines for Soil Washing. Interstate Technology and Regulatory Council. [ | + | Improving the efficiency and reducing the cost of excavation, pump-and-treat, electrokinetics, and phytoextraction remains an important pursuit. Sometimes, the technologies in Table 1 can be combined to reduce costs. For excavation, the primary focus has been on reducing the volume of contaminated soil needing special disposal. This can involve soil washing<ref>ITRC, 1997. Technical and Regulatory Guidelines for Soil Washing. Interstate Technology and Regulatory Council. [[Media:ITRC-1997-Tech_%26_Reg_Guidelines_for_Soil_Washing.pdf|Report pdf]]</ref><ref>Dermont, G., Bergeron, M., Mercier, G. and Richer-Laflèche, M., 2008. Soil washing for metal removal: a review of physical/chemical technologies and field applications. Journal of Hazardous Materials, 152(1), 1-31. [http://dx.doi.org/10.1016/j.jhazmat.2007.10.043 doi: 10.1016/j.jhazmat.2007.10.043]</ref> where water-based solutions of chemicals are used to remove metals from excavated soils, though electrokinetics and phytoextraction can also be used to clean contaminated soil piles. Soil flushing involves injecting reagents into the subsurface that mobilize the metal contaminants to make them more available for removal by pump-and-treat, electrokinetics, and phytoextraction<ref>Roote, D.S., 1997. In Situ Flushing. TO-97-02. Ground-water remediation technologies analysis center.[[Media:Roote-2008-In_Situ_Flushing.pdf|Report pdf]]</ref><ref>Leštan, D., Luo, C.L. and Li, X.D., 2008. The use of chelating agents in the remediation of metal-contaminated soils: a review. Environmental Pollution,153(1), 3-13. [http://dx.doi.org/10.1016/j.envpol.2007.11.015 doi: 10.1016/j.envpol.2007.11.015]</ref>. |
The most common approaches for managing soils contamination at smaller metals sites are excavation and phytoextraction. Pump-and-treat systems are widely used for metal and metalloid contaminated groundwater. Electrokinetic projects for remediating metals sites are now relatively rare with few applications after the mid 2000s. | The most common approaches for managing soils contamination at smaller metals sites are excavation and phytoextraction. Pump-and-treat systems are widely used for metal and metalloid contaminated groundwater. Electrokinetic projects for remediating metals sites are now relatively rare with few applications after the mid 2000s. | ||
==In Situ Remediation== | ==In Situ Remediation== | ||
| − | Issues with the access, cost, and efficiency of metals and metalloids removal from the subsurface have driven efforts to develop methods that rely solely on in situ reactions to minimize the movement of these contaminants towards points of exposure. These efforts can range from monitored natural attenuation of metals<ref name="EPA2015">U.S. Environmental Protection Agency, 2015. Use of Monitored Natural Attenuation for Inorganic contaminants in Groundwater at Superfund Sites. Directive 9283.1-36, Office of Solid Waste and Emergency Response. [ | + | Issues with the access, cost, and efficiency of metals and metalloids removal from the subsurface have driven efforts to develop methods that rely solely on in situ reactions to minimize the movement of these contaminants towards points of exposure. These efforts can range from monitored natural attenuation of metals<ref name="EPA2015">U.S. Environmental Protection Agency, 2015. Use of Monitored Natural Attenuation for Inorganic contaminants in Groundwater at Superfund Sites. Directive 9283.1-36, Office of Solid Waste and Emergency Response. [[Media:MNA-Guidance-2015.pdf|Report pdf]]</ref> that relies solely on natural processes to engineered treatments deployed in the subsurface. Examples of engineered treatments include: |
| − | *Adding an electron donor, such as lactate, to biologically turn the groundwater anaerobic, thereby precipitating or increasing the sorption metals such as chromium, uranium, cadmium, copper, lead, and zinc in groundwater. Sometimes, sulfate is also added with the electron donor so this reaction will generate hydrogen sulfide, which can also precipitate these metals<ref name = "NABIR2003">NABIR, 2003. Bioremediation of metals and radionuclides – what it is and how it works. LBNL-42595, Lawrence Berkeley National Laboratory for the Natural and Accelerated Bioremediation Research Program, Office of Science, U.S. Department of Energy [ | + | *Adding an electron donor, such as lactate, to biologically turn the groundwater anaerobic, thereby precipitating or increasing the sorption metals such as chromium, uranium, cadmium, copper, lead, and zinc in groundwater. Sometimes, sulfate is also added with the electron donor so this reaction will generate hydrogen sulfide, which can also precipitate these metals<ref name = "NABIR2003">NABIR, 2003. Bioremediation of metals and radionuclides – what it is and how it works. LBNL-42595, Lawrence Berkeley National Laboratory for the Natural and Accelerated Bioremediation Research Program, Office of Science, U.S. Department of Energy [[Media:NABIR-2003-Bioremediation_of_Metals_and_Radionuclides.pdf|Report pdf]]</ref> |
*Adding oxygen to turn groundwater more aerobic, thereby reducing the mobility of arsenic<ref name="Hashim2011"/> | *Adding oxygen to turn groundwater more aerobic, thereby reducing the mobility of arsenic<ref name="Hashim2011"/> | ||
| Line 65: | Line 65: | ||
#Chemical barriers to contaminant migration. | #Chemical barriers to contaminant migration. | ||
| − | Physical barriers include subsurface walls that block or redirect groundwater flow and methods of trapping contaminants in engineered low permeability zones. An example is a slurry wall where a low-permeability benonite material is emplaced in a trench around groundwater source zone to reduce the flow through this source zone<ref name= "Pearlman1999">Pearlman, L., 1999. Subsurface containment and monitoring systems: Barriers and beyond. National Network of Environmental Management Studies Fellow for US Environmental Protection Agency, pgs. 1-61. [ | + | Physical barriers include subsurface walls that block or redirect groundwater flow and methods of trapping contaminants in engineered low permeability zones. An example is a slurry wall where a low-permeability benonite material is emplaced in a trench around groundwater source zone to reduce the flow through this source zone<ref name= "Pearlman1999">Pearlman, L., 1999. Subsurface containment and monitoring systems: Barriers and beyond. National Network of Environmental Management Studies Fellow for US Environmental Protection Agency, pgs. 1-61. [[Media:Pearlman-1999-Barriers_and_Beyond.pdf|Report pdf]]</ref>. Chemical barriers involve placement of reagents in the subsurface that react with contaminants to minimize their mobility in groundwater. The reagents either react directly with the contaminants or change the geochemistry of the subsurface in a way that reduces contaminant mobility. An example of this approach is construction of a permeable reactive barrier filled with [[Zerovalent Iron (ZVI) (Chemical Reduction - ISCR) (Chemical Reduction - ISCR) (Chemical Reduction - ISCR) | zerovalent iron]] that chemically reduces soluble uranium(VI) entering the barrier to uranium(IV) that is removed from groundwater by precipitation<ref name= "Naftz2002">Naftz, D., Morrison, S.J., Fuller, C.C. and Davis, J.A. eds., 2002. Handbook of groundwater remediation using permeable reactive barriers: applications to radionuclides, trace metals, and nutrients. Academic Press. ISBN 9780080533056</ref><ref name = "ITRC2011PRB">ITRC, 2011. Permeable Reactive Barrier: Technology Update. Interstate Technology and Regulatory Council. [[Media:ITRC-2011-PRB_Tech_Update.pdf|Report pdf]]</ref>. Treatment by chemical barriers is often referred to as enhanced attenuation because the purpose is to increase the attenuation capacity of the aquifer. Combinations of physical and chemical barriers can also be used. For example, “funnel-and-gate” systems use subsurface physical barriers to direct contaminated groundwater through “gates” where reagents are deployed to react with the contaminants. |
===Design Considerations=== | ===Design Considerations=== | ||
| Line 93: | Line 93: | ||
| Subsurface Barriers|| Block contaminated groundwater flow by sheet piling, cementitious barriers, slurry walls, frozen barriers, etc.|| Immediate effect; relatively simple to design; widely available.|| Depth and geology limitations; difficult to predict long-term degradation; requires mitigation of contaminated groundwater bypassing wall (flow around, beneath, over barrier.)|| Pearlman, 1999 <ref name= "Pearlman1999"/>; Evanko and Dzombak, 1997<ref name="Evanko1997"/>; NAS, 1999<ref name= "NAS1999"/> | | Subsurface Barriers|| Block contaminated groundwater flow by sheet piling, cementitious barriers, slurry walls, frozen barriers, etc.|| Immediate effect; relatively simple to design; widely available.|| Depth and geology limitations; difficult to predict long-term degradation; requires mitigation of contaminated groundwater bypassing wall (flow around, beneath, over barrier.)|| Pearlman, 1999 <ref name= "Pearlman1999"/>; Evanko and Dzombak, 1997<ref name="Evanko1997"/>; NAS, 1999<ref name= "NAS1999"/> | ||
|- | |- | ||
| − | | In Situ Solidification|| Trap contaminants in engineered low permeability block.|| Immediate effect; relatively easy to design; widely available.|| Depth and geology limitations; difficult to predict long-term degradation; rarely used.|| Bates and Hill, 2015<ref>Bates, E. and C. Hill, 2015. Stabilization and Solidification of Contaminated Soil and Waste: A Manual of Practice. [ | + | | In Situ Solidification|| Trap contaminants in engineered low permeability block.|| Immediate effect; relatively easy to design; widely available.|| Depth and geology limitations; difficult to predict long-term degradation; rarely used.|| Bates and Hill, 2015<ref>Bates, E. and C. Hill, 2015. Stabilization and Solidification of Contaminated Soil and Waste: A Manual of Practice. [[Media:Bates-2015-Stabilization_and_Solidification_of_Contaminated_Soil_and_Waste.pdf|Report pdf]]</ref>; Evanko and Dzombak, 1997<ref name="Evanko1997"/>; NAS, 1999<ref name= "NAS1999"/> |
|- | |- | ||
| − | | In Situ Vitrification|| Trap contaminants in a solid vitreous mass by heating subsurface solids to melting or fusing point.|| Immediate effect.|| Depth and geology limitations; difficult to predict long-term degradation; specific soil conditions required; rarely used.|| U.S. EPA, 1994<ref>U.S. Environmental Protection Agency (USEPA), 1994. Engineering Bulletin – In Situ Vitrification. EPA/540/S-94/504. Office of Emergency and Remedial Response. [ | + | | In Situ Vitrification|| Trap contaminants in a solid vitreous mass by heating subsurface solids to melting or fusing point.|| Immediate effect.|| Depth and geology limitations; difficult to predict long-term degradation; specific soil conditions required; rarely used.|| U.S. EPA, 1994<ref>U.S. Environmental Protection Agency (USEPA), 1994. Engineering Bulletin – In Situ Vitrification. EPA/540/S-94/504. Office of Emergency and Remedial Response. [[Media:USEPA-1994-In_Situ_Vitrification.pdf|Report pdf]]</ref>; Evanko and Dzombak, 1997<ref name="Evanko1997"/>; NAS, 1999<ref name= "NAS1999"/> |
|- | |- | ||
| − | | Permeable Reactive Barriers (PRB)|| Typically (definition varies) a trench or constructed subsurface wall that contains a reagent that allows the passage of groundwater while trapping contaminants.|| Relatively easy to design and construct; can use wide variety of reagents in various combinations for treating multiple contaminants; can be excavated when no longer needed. || Depth and geology limitations; reactive media is subject to deactivation by coating of reaction products; intra-barrier monitoring possible.|| Naftz, et al., 2002<ref name= "Naftz2002"/>; ITRC, 1999<ref>ITRC, 1999. Regulatory Guidance for Permeable Reactive Barriers Designed to Remediate Inorganic and Radionuclide Contamination. Interstate Technology and Regulatory Council. [ | + | | Permeable Reactive Barriers (PRB)|| Typically (definition varies) a trench or constructed subsurface wall that contains a reagent that allows the passage of groundwater while trapping contaminants.|| Relatively easy to design and construct; can use wide variety of reagents in various combinations for treating multiple contaminants; can be excavated when no longer needed. || Depth and geology limitations; reactive media is subject to deactivation by coating of reaction products; intra-barrier monitoring possible.|| Naftz, et al., 2002<ref name= "Naftz2002"/>; ITRC, 1999<ref>ITRC, 1999. Regulatory Guidance for Permeable Reactive Barriers Designed to Remediate Inorganic and Radionuclide Contamination. Interstate Technology and Regulatory Council. [[Media:ITRC-1999-Permeable_Reactive_Barriers_for_Inorganics_and_Radionuclides.pdf|Report pdf]]</ref>; ITRC, 2011<ref name = "ITRC2011PRB"/>; Evanko and Dzombak, 1997<ref name="Evanko1997"/>; NAS, 1999<ref name= "NAS1999"/>; Mulligan et al., 2001<ref name="Mulligan2001"/> |
|- | |- | ||
| Funnel-and-Gate|| Combination of physical and chemical barriers with PRB or in situ treatment zone at gate.|| Limits volume of treatment zone; useful for a really extensive plumes.|| Depth and geology limitations; predicting long-term degradation; rarely used in recent years.|| Sedivy et al., 1999<ref>Sedivy, R.A., Shafer, J.M. and Bilbrey, L.C., 1999. Design screening tools for passive funnel and gate systems. Groundwater Monitoring & Remediation, 19(1), 125-133. [http://dx.doi.org/10.1111/j.1745-6592.1999.tb00195.x doi: 10.1111/j.1745-6592.1999.tb00195.x]</ref> | | Funnel-and-Gate|| Combination of physical and chemical barriers with PRB or in situ treatment zone at gate.|| Limits volume of treatment zone; useful for a really extensive plumes.|| Depth and geology limitations; predicting long-term degradation; rarely used in recent years.|| Sedivy et al., 1999<ref>Sedivy, R.A., Shafer, J.M. and Bilbrey, L.C., 1999. Design screening tools for passive funnel and gate systems. Groundwater Monitoring & Remediation, 19(1), 125-133. [http://dx.doi.org/10.1111/j.1745-6592.1999.tb00195.x doi: 10.1111/j.1745-6592.1999.tb00195.x]</ref> | ||
Revision as of 13:48, 3 May 2018
There are many sites where soil and/or groundwater are contaminated with metals and metalloids (collectively referred to as “metals” henceforth) that require some type of response. The most commonly occurring metals at Superfund sites are lead, chromium, arsenic, zinc, cadmium, copper, and mercury. Many remediation technologies that are used at sites with organic contaminants can be also be used at sites with metals contamination with one major caveat: metals are not destroyed by chemical or biological degradation. At smaller sites, metal contaminants in soils are commonly treated by excavation. In groundwater, metals plumes are treated by pump-and-treat systems, monitored natural attenuation of metals, or remediated by changing the groundwater geochemistry to immobilize the metal contaminants to prevent migration to receptors.
Related Article(s):
- Metal and Metalloid Contaminants
- Metals and Metalloids - Mobility in Groundwater
- Monitored Natural Attenuation (MNA) of Metal and Metalloids
CONTRIBUTOR(S): Dr. Miles Denham and Dr. Charles Newell, P.E.
Key Resource(s):
- Groundwater and Soil Cleanup: Improving Management of Persistent Contaminants[2]
Introduction
Engineered remediation of metal and metalloid contaminants in groundwater can be accomplished by (a) removal of the contaminants from the subsurface or (b) treating them in situ to reduce their mobility or concentration to levels considered safe to human health or the environment. In situ treatment means the contaminant metals are left in the subsurface. Therefore, it must be demonstrated that the rate of release of the contaminants from the treatment zone will be low and stable enough that the contaminants will pose minimal risk over a long period of time. Excellent reviews of technologies for remediation of metal and metalloid contaminated groundwater are available such as:
- Remediation of Metals Contaminated Soils and Groundwater (1997)[1]: Describes the sources, fate, transport, and influence of soil properties on mobility and then discusses general remediation approaches followed by several commercial processes.
- Groundwater and Soil Cleanup: Improving Management of Persistent Contaminants (1999)[2]: Focuses on both mobilization technologies (in order to move metals to a location where it can be treated) and immobilization technologies (in order to stabilize metals in place and prevent further spreading).
- Remediation Technologies for Metal-Contaminated Soils and Groundwater; an Evaluation (2001)[4]: Explains how remediation approach depends on site characteristics, concentration, pollutants types, and the final use of the soil or groundwater. Key approaches include isolation, immobilization, toxicity reduction, physical separation, and extraction.
- Remediation Technologies for Heavy Metal Contaminated Groundwater (2011)[5]: Describes “Thirty-five approaches for groundwater treatment have been reviewed and classified under three large categories viz chemical, biochemical/biological/biosorption and physico-chemical treatment processes.”
Subsurface Removal of Contaminants
The advantage of remediation technologies that remove metal and metalloid contaminants from the subsurface is that, when remediation is complete, the risk is eliminated. Excavation and pump-and-treat are also widely available and accepted by regulators and stakeholders. However, the primary disadvantage of technologies that remove contaminants from the subsurface is that they create contaminated solid waste that must be disposed. Additional issues are worker exposure, prolonged disturbance of the surface environment, and long-term maintenance costs for approaches relying on treating groundwater at the surface. Overall, there is a large range of contaminant removal technologies each with advantages and disadvantages (Table 1).
| Technology (Target Media) | Description | Advantages | Disadvantages | Reference(s) |
|---|---|---|---|---|
| Excavation (Contaminated soils) | Removal of contaminated soils in unsaturated zone to eliminate secondary source of contamination. | Widely available and accepted. | Creates large amount of solid waste; not feasible in some geology; expense increases significantly for excavation below the water table. | Post et al., 2013[6] |
| Pump-and-Treat (Groundwater plumes) | Extraction of contaminated groundwater by well network for treatment at surface. | Widely available and accepted. | Surface treatment system can be expensive; can require long-time frames to reach remedial goals because of sorption of metals and metalloids. | Mackay and Cherry, 1989[7]; Mercer et al., 1990[8] |
| Electrokinetics
(Contaminated fine grained material such as clays) |
Deployment of electrodes in subsurface to create an electrical field that drives contaminants to the electrodes. | Effective in clay-rich aquifers; potential for less solid waste than excavation or pump-and-treat. | Rarely used; increase of pH near cathode causing precipitation of metal salts; efficiency decreases outside of specific aquifer and contamination conditions. | Van Cauwenberghe, 1997[9]; Virkutyte et al., 2002[10]; Vocciante et al., 2016[11] |
| Phytoextraction
(Mostly contaminated soils, sometimes groundwater) |
Use of plants to extract contaminants from the subsurface. | Only periodic maintenance (harvesting and processing of plants or plant detritus) once plants are established. | Need long-term access to treat soils; difficult to treat deep groundwater; requires hyperaccumulating plants that may not exist for metals that are not essential nutrients. | Pivetz, 2001[12]; McGrath and Zhao, 2003[13]; Sheoran et al., 2016[14] |
Improving the efficiency and reducing the cost of excavation, pump-and-treat, electrokinetics, and phytoextraction remains an important pursuit. Sometimes, the technologies in Table 1 can be combined to reduce costs. For excavation, the primary focus has been on reducing the volume of contaminated soil needing special disposal. This can involve soil washing[15][16] where water-based solutions of chemicals are used to remove metals from excavated soils, though electrokinetics and phytoextraction can also be used to clean contaminated soil piles. Soil flushing involves injecting reagents into the subsurface that mobilize the metal contaminants to make them more available for removal by pump-and-treat, electrokinetics, and phytoextraction[17][18].
The most common approaches for managing soils contamination at smaller metals sites are excavation and phytoextraction. Pump-and-treat systems are widely used for metal and metalloid contaminated groundwater. Electrokinetic projects for remediating metals sites are now relatively rare with few applications after the mid 2000s.
In Situ Remediation
Issues with the access, cost, and efficiency of metals and metalloids removal from the subsurface have driven efforts to develop methods that rely solely on in situ reactions to minimize the movement of these contaminants towards points of exposure. These efforts can range from monitored natural attenuation of metals[19] that relies solely on natural processes to engineered treatments deployed in the subsurface. Examples of engineered treatments include:
- Adding an electron donor, such as lactate, to biologically turn the groundwater anaerobic, thereby precipitating or increasing the sorption metals such as chromium, uranium, cadmium, copper, lead, and zinc in groundwater. Sometimes, sulfate is also added with the electron donor so this reaction will generate hydrogen sulfide, which can also precipitate these metals[20]
- Adding oxygen to turn groundwater more aerobic, thereby reducing the mobility of arsenic[5]
In situ remediation requires strong evidence that remaining, treated contaminants will not be a threat for long time periods. For example, when mobile Chrome(VI) (oxidation state of six) is converted to immobile Chrome(III) by adding electron donor, it does not reoxidize back to Chrome(VI) except under unique conditions (e.g., high manganese concentrations)[3]. The strength of the evidence for effective treatment usually depends on the toxicity of the contaminant and the length of time it will take the contaminant to reach receptors at hazardous concentrations. In situ treatments are not designed to eliminate the flux of contaminants toward receptors, but rather to keep the flux below levels that are a threat.
In situ remediation can be divided into two broad categories:
- Physical barriers to contaminant migration, and
- Chemical barriers to contaminant migration.
Physical barriers include subsurface walls that block or redirect groundwater flow and methods of trapping contaminants in engineered low permeability zones. An example is a slurry wall where a low-permeability benonite material is emplaced in a trench around groundwater source zone to reduce the flow through this source zone[21]. Chemical barriers involve placement of reagents in the subsurface that react with contaminants to minimize their mobility in groundwater. The reagents either react directly with the contaminants or change the geochemistry of the subsurface in a way that reduces contaminant mobility. An example of this approach is construction of a permeable reactive barrier filled with zerovalent iron that chemically reduces soluble uranium(VI) entering the barrier to uranium(IV) that is removed from groundwater by precipitation[22][23]. Treatment by chemical barriers is often referred to as enhanced attenuation because the purpose is to increase the attenuation capacity of the aquifer. Combinations of physical and chemical barriers can also be used. For example, “funnel-and-gate” systems use subsurface physical barriers to direct contaminated groundwater through “gates” where reagents are deployed to react with the contaminants.
Design Considerations
Engineered treatment of contaminants in situ requires careful consideration of groundwater flow to ensure that treatments are placed in the optimal locations and orientations. In addition, the effects of the treatment on groundwater flow must be considered. Physical barriers redirect groundwater flow by design. In the case of subsurface barriers designed to block groundwater flow, the groundwater will “pile” up at the barrier, flow around the barrier, and/or flow beneath the barrier and designs must incorporate these consequences. Likewise, chemical barriers can sometimes reduce permeability forcing contaminated groundwater around the treated zone.
The design and limitations of physical barriers depend primarily on the characteristics of the site rather than the contaminants present. For example, if there is hard crystalline rock between the surface and the contamination, it may be difficult to install a physical barrier. In contrast, site characteristics influence design of chemical barriers or enhanced attenuation, but the primary consideration is the contaminants present.
Enhanced Attenuation Remedies
Paraphrasing and rearranging the four tiers of evidence required by the U.S. Environmental Protection Agency[19] to demonstrate monitored natural attenuation (MNA) for inorganic compounds provides a general guide to designing an enhanced attenuation remediation:
- Decide on a mechanism/reagent and determine the rate of attenuation necessary.
- Determine the capacity needed for long-term attenuation of the contaminant plume.
- Demonstrate long-term stability of the attenuated contaminants in bench and field-scale tests.
- Design a performance monitoring program and identify mitigation strategies for undesirable collateral effects.
The U.S. Department of Energy has produced an aid to design enhanced attenuation remedies that categorizes sites and contaminants into groups with similar characteristics[3] (see Monitored Natural Attenuation (MNA) of Metal and Metalloids). Sites and contaminants within each group can be treated with generally similar approaches and mechanisms. Demonstrating long-term stability of attenuated contaminants is the most difficult challenge to deploying enhanced attenuation remedies. Contaminant plumes are dynamic systems and conditions today may be very different from conditions in the future. In general, the geochemical conditions of a waste site evolve from the contaminated conditions back toward natural conditions. Demonstration of long-term effectiveness for a remedy with a primary attenuation mechanism that is consistent with this evolution is more convincing than for a remedy with a primary attenuation mechanism that is likely to be reversed by the natural evolution of the site. Imagine an “Attenuation Conceptual Model” that can help guide if, when, and where metals MNA, enhanced attenuation, or more aggressive remediation is required (Fig. 1)[3]. Sometimes reactive transport modeling can help understand the geochemistry of groundwater at complicated metals sites.
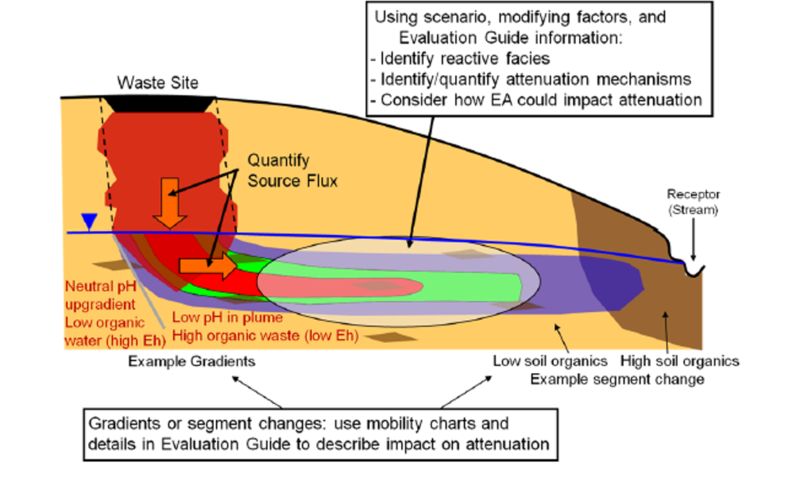
There are numerous in situ treatment technologies for metals and metalloid contaminants (Table 2). For enhanced attenuation, there is a wide spectrum of reagents that may be used that is too extensive to cover here. Hence, we only outline the general mechanisms in our compilation here.
| Technology | Description | Advantages | Disadvantages | Reference(s) |
|---|---|---|---|---|
| Capping | Construction of low permeability cap or cover to prevent infiltration through metal contaminated soil, thereby reducing mobility. | Inexpensive; provides long-term solution; proven technology; very common solution. | Does not reduce toxicity or volume of metals contaminants. | Evanko and Dzombak, 1997[1] |
| Subsurface Barriers | Block contaminated groundwater flow by sheet piling, cementitious barriers, slurry walls, frozen barriers, etc. | Immediate effect; relatively simple to design; widely available. | Depth and geology limitations; difficult to predict long-term degradation; requires mitigation of contaminated groundwater bypassing wall (flow around, beneath, over barrier.) | Pearlman, 1999 [21]; Evanko and Dzombak, 1997[1]; NAS, 1999[2] |
| In Situ Solidification | Trap contaminants in engineered low permeability block. | Immediate effect; relatively easy to design; widely available. | Depth and geology limitations; difficult to predict long-term degradation; rarely used. | Bates and Hill, 2015[24]; Evanko and Dzombak, 1997[1]; NAS, 1999[2] |
| In Situ Vitrification | Trap contaminants in a solid vitreous mass by heating subsurface solids to melting or fusing point. | Immediate effect. | Depth and geology limitations; difficult to predict long-term degradation; specific soil conditions required; rarely used. | U.S. EPA, 1994[25]; Evanko and Dzombak, 1997[1]; NAS, 1999[2] |
| Permeable Reactive Barriers (PRB) | Typically (definition varies) a trench or constructed subsurface wall that contains a reagent that allows the passage of groundwater while trapping contaminants. | Relatively easy to design and construct; can use wide variety of reagents in various combinations for treating multiple contaminants; can be excavated when no longer needed. | Depth and geology limitations; reactive media is subject to deactivation by coating of reaction products; intra-barrier monitoring possible. | Naftz, et al., 2002[22]; ITRC, 1999[26]; ITRC, 2011[23]; Evanko and Dzombak, 1997[1]; NAS, 1999[2]; Mulligan et al., 2001[4] |
| Funnel-and-Gate | Combination of physical and chemical barriers with PRB or in situ treatment zone at gate. | Limits volume of treatment zone; useful for a really extensive plumes. | Depth and geology limitations; predicting long-term degradation; rarely used in recent years. | Sedivy et al., 1999[27] |
| In Situ Treatment Zones - Abiotic | Reagents such as zero valent iron are injected into subsurface to induce reactions that limit contaminant mobility (i.e., enhanced sorption, precipitation, ion exchange). | Minimally invasive (only injection wells required); useful for deep plumes and in a wide variety of geologic settings; useful when subsurface infrastructure is present; commonly used. | Uncertainty in distribution of reagent; number of injection wells required can offset advantage of being minimally invasive; may require repeated treatments. | Hashim et al., 2011[5]; NAS, 1999[2] |
| In Situ Treatment Zones – Biotic (Bioremediation) | Reagents such as lactate, vegetable oil, and mulch (and sometimes microbes) injected into subsurface to stimulate reduction reactions that limit mobility of redox sensitive contaminants or those that precipitate as metal sulfides (biosorption technologies have been tested, but not in wide use). | Minimally invasive (only injection wells required); useful for deep plumes and in a wide variety of geologic settings; useful when subsurface infrastructure is present; commonly used. | Only useful for contaminants that are less mobile in a reduced oxidation state or as sulfides; subject to reversal in aerobic aquifers (less so for Cr(VI)); requires secondary attenuation mechanism if contaminant is re-oxidized. | NABIR, 2003[20]; Hashim et al., 2011[5] |
References
- ^ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Evanko, C.R. and Dzombak, D.A., 1997. Remediation of metals-contaminated soils and groundwater. TE-97-01. Ground-water remediation technologies analysis center. Report pdf
- ^ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 National Research Council, 1999. Groundwater and soil cleanup: Improving management of persistent contaminants. National Academies Press.
- ^ 3.0 3.1 3.2 3.3 3.4 Truex, M., Brady, P., Newell, C.J., Rysz, M., Denham, M., Vangelas, K. 2011. The scenarios approach to attenuation-based remedies for inorganic and radionuclide contaminants. Savannah-River National Laboratory U.S. Department of Energy. Report pdf
- ^ 4.0 4.1 Mulligan, C.N., Yong, R.N. and Gibbs, B.F., 2001. Remediation technologies for metal-contaminated soils and groundwater: an evaluation. Engineering Geology, 60(1), 193-207. doi: 10.1016/S0013-7952(00)00101-0
- ^ 5.0 5.1 5.2 5.3 Hashim, M.A., Mukhopadhyay, S., Sahu, J.N. and Sengupta, B., 2011. Remediation technologies for heavy metal contaminated groundwater. Journal of Environmental Management, 92(10), 2355-2388. doi:10.1016/j.jenvman.2011.06.009
- ^ Post, T.C., Strom, D. and Beulow, L., 2013, July. The 100-C-7 Remediation Project. An Overview of One of DOE's Largest Remediation Projects-13260. WM Symposia, Tempe, AZ, 85282 (United States). Report pdf
- ^ Mackay, D.M. and Cherry, J.A., 1989. Groundwater contamination: Pump-and-treat remediation. Environmental Science & Technology, 23(6), 630-636. doi: 10.1021/es00064a001
- ^ Mercer, J.W., Skipp, D.C. and Giffin, D., 1990. Basics of pump-and-treat ground-water remediation technology, pgs. 1-66. EPA-600/8-90/003. Robert S. Kerr Environmental Research Laboratory, Office of Research and Development, US Environmental Protection Agency. Report pdf
- ^ Van Cauwenberghe, L., 1997. Electrokinetics. TO-97-03. Ground-water remediation technologies analysis center. Report pdf
- ^ Virkutyte, J., Sillanpää, M. and Latostenmaa, P., 2002. Electrokinetic soil remediation—critical overview. Science of the Total Environment, 289(1), 97-121. doi: 10.1016/S0048-9697(01)01027-0
- ^ Vocciante, M., Caretta, A., Bua, L., Bagatin, R. and Ferro, S., 2016. Enhancements in ElectroKinetic Remediation Technology: Environmental assessment in comparison with other configurations and consolidated solutions. Chemical Engineering Journal, 289, 123-134. doi:10.1016/j.cej.2015.12.065
- ^ Pivetz, B.E., 2001. Phytoremediation of Contaminated Soil and Ground Water at Hazardous Waste Sites. EPA/540/S-01/500. US Environmental Protection Agency. Report pdf
- ^ McGrath, S.P. and Zhao, F.J., 2003. Phytoextraction of metals and metalloids from contaminated soils. Current Opinion in Biotechnology, 14(3), 277-282. doi: 10.1016/S0958-1669(03)00060-0
- ^ Sheoran, V., Sheoran, A.S. and Poonia, P., 2016. Factors affecting phytoextraction: A review. Pedosphere, 26(2), 148-166. doi: 10.1016/S1002-0160(15)60032-7
- ^ ITRC, 1997. Technical and Regulatory Guidelines for Soil Washing. Interstate Technology and Regulatory Council. Report pdf
- ^ Dermont, G., Bergeron, M., Mercier, G. and Richer-Laflèche, M., 2008. Soil washing for metal removal: a review of physical/chemical technologies and field applications. Journal of Hazardous Materials, 152(1), 1-31. doi: 10.1016/j.jhazmat.2007.10.043
- ^ Roote, D.S., 1997. In Situ Flushing. TO-97-02. Ground-water remediation technologies analysis center.Report pdf
- ^ Leštan, D., Luo, C.L. and Li, X.D., 2008. The use of chelating agents in the remediation of metal-contaminated soils: a review. Environmental Pollution,153(1), 3-13. doi: 10.1016/j.envpol.2007.11.015
- ^ 19.0 19.1 U.S. Environmental Protection Agency, 2015. Use of Monitored Natural Attenuation for Inorganic contaminants in Groundwater at Superfund Sites. Directive 9283.1-36, Office of Solid Waste and Emergency Response. Report pdf
- ^ 20.0 20.1 NABIR, 2003. Bioremediation of metals and radionuclides – what it is and how it works. LBNL-42595, Lawrence Berkeley National Laboratory for the Natural and Accelerated Bioremediation Research Program, Office of Science, U.S. Department of Energy Report pdf
- ^ 21.0 21.1 Pearlman, L., 1999. Subsurface containment and monitoring systems: Barriers and beyond. National Network of Environmental Management Studies Fellow for US Environmental Protection Agency, pgs. 1-61. Report pdf
- ^ 22.0 22.1 Naftz, D., Morrison, S.J., Fuller, C.C. and Davis, J.A. eds., 2002. Handbook of groundwater remediation using permeable reactive barriers: applications to radionuclides, trace metals, and nutrients. Academic Press. ISBN 9780080533056
- ^ 23.0 23.1 ITRC, 2011. Permeable Reactive Barrier: Technology Update. Interstate Technology and Regulatory Council. Report pdf
- ^ Bates, E. and C. Hill, 2015. Stabilization and Solidification of Contaminated Soil and Waste: A Manual of Practice. Report pdf
- ^ U.S. Environmental Protection Agency (USEPA), 1994. Engineering Bulletin – In Situ Vitrification. EPA/540/S-94/504. Office of Emergency and Remedial Response. Report pdf
- ^ ITRC, 1999. Regulatory Guidance for Permeable Reactive Barriers Designed to Remediate Inorganic and Radionuclide Contamination. Interstate Technology and Regulatory Council. Report pdf
- ^ Sedivy, R.A., Shafer, J.M. and Bilbrey, L.C., 1999. Design screening tools for passive funnel and gate systems. Groundwater Monitoring & Remediation, 19(1), 125-133. doi: 10.1111/j.1745-6592.1999.tb00195.x