Difference between revisions of "User:Jhurley/sandbox"
(→Carbonaceous Reductants) |
(→Carbonaceous Reductants) |
||
| Line 54: | Line 54: | ||
! AHQS<sup>-</sup> | ! AHQS<sup>-</sup> | ||
|- | |- | ||
| − | | Nitrobenzene (NB) || -0.485 || 0.380 || -1.102 || 2.050 || 3.060 | + | | Nitrobenzene (NB) || -0.485<ref name="Schwarzenbach1990"/> || 0.380<ref name="Schwarzenbach1990"/> || -1.102<ref name="Schwarzenbach1990"/> || 2.050 || 3.060 |
|- | |- | ||
| − | | 2-nitrotoluene (2-NT) || -0.590 || -1.432 || -2.523 || 0.775 || | + | | 2-nitrotoluene (2-NT) || -0.590<ref name="Schwarzenbach1990"/> || -1.432<ref name="Schwarzenbach1990"/> || -2.523<ref name="Schwarzenbach1990"/> || 0.775 || |
|- | |- | ||
| − | | 3-nitrotoluene (3-NT) || -0.475 || 0.462 || -0.921 || || | + | | 3-nitrotoluene (3-NT) || -0.475<ref name="Schwarzenbach1990"/> || 0.462<ref name="Schwarzenbach1990"/> || -0.921<ref name="Schwarzenbach1990"/> || || |
|- | |- | ||
| − | | 4-nitrotoluene (4-NT) || -0.500 || 0.041 || -1.292 || 1.822 || 2.610 | + | | 4-nitrotoluene (4-NT) || -0.500<ref name="Schwarzenbach1990"/> || 0.041<ref name="Schwarzenbach1990"/> || -1.292<ref name="Schwarzenbach1990"/> || 1.822 || 2.610 |
|- | |- | ||
| − | | 2-chloronitrobenzene (2-ClNB) || -0.485 || 0.342 || -0.824 ||2.412 || | + | | 2-chloronitrobenzene (2-ClNB) || -0.485<ref name="Schwarzenbach1990"/> || 0.342<ref name="Schwarzenbach1990"/> || -0.824<ref name="Schwarzenbach1990"/> ||2.412 || |
|- | |- | ||
| − | | 3-chloronitrobenzene (3-ClNB) || -0.405 || 1.491 || 0.114 || || | + | | 3-chloronitrobenzene (3-ClNB) || -0.405<ref name="Schwarzenbach1990"/> || 1.491<ref name="Schwarzenbach1990"/> || 0.114<ref name="Schwarzenbach1990"/> || || |
|- | |- | ||
| − | | 4-chloronitrobenzene (4-ClNB) || -0.450 || 1.041 || -0.301 || 2.988 || | + | | 4-chloronitrobenzene (4-ClNB) || -0.450<ref name="Schwarzenbach1990"/> || 1.041<ref name="Schwarzenbach1990"/> || -0.301<ref name="Schwarzenbach1990"/> || 2.988 || |
|- | |- | ||
| − | | 2-acetylnitrobenzene (2-AcNB) || -0.470 || 0.519 || -0.456 || || | + | | 2-acetylnitrobenzene (2-AcNB) || -0.470<ref name="Schwarzenbach1990"/> || 0.519<ref name="Schwarzenbach1990"/> || -0.456<ref name="Schwarzenbach1990"/> || || |
|- | |- | ||
| − | | 3-acetylnitrobenzene (3-AcNB) || -0.405 || 1.663 || 0.398 || || | + | | 3-acetylnitrobenzene (3-AcNB) || -0.405<ref name="Schwarzenbach1990"/> || 1.663<ref name="Schwarzenbach1990"/> || 0.398<ref name="Schwarzenbach1990"/> || || |
|- | |- | ||
| − | | 4-acetylnitrobenzene (4-AcNB) || -0.360 || 2.519 || 1.477 || || | + | | 4-acetylnitrobenzene (4-AcNB) || -0.360<ref name="Schwarzenbach1990"/> || 2.519<ref name="Schwarzenbach1990"/> || 1.477<ref name="Schwarzenbach1990"/> || || |
|- | |- | ||
| − | | 2-nitrophenol (2-NP) || || 0.568 (0.079) || || || | + | | 2-nitrophenol (2-NP) || || 0.568 (0.079)<ref name="Schwarzenbach1990"/> || || || |
|- | |- | ||
| − | | 4-nitrophenol (4-NP) || || -0.699 (-1.301) || || || | + | | 4-nitrophenol (4-NP) || || -0.699 (-1.301)<ref name="Schwarzenbach1990"/> || || || |
|- | |- | ||
| − | | 4-methyl-2-nitrophenol (4-Me-2-NP) || || 0.748 (0.176) || || || | + | | 4-methyl-2-nitrophenol (4-Me-2-NP) || || 0.748 (0.176)<ref name="Schwarzenbach1990"/> || || || |
|- | |- | ||
| − | | 4-chloro-2-nitrophenol (4-Cl-2-NP) || || 1.602 (1.114) || || || | + | | 4-chloro-2-nitrophenol (4-Cl-2-NP) || || 1.602 (1.114)<ref name="Schwarzenbach1990"/> || || || |
|- | |- | ||
| − | | 5-fluoro-2-nitrophenol (5-Cl-2-NP) || || 0.447 (-0.155) || || || | + | | 5-fluoro-2-nitrophenol (5-Cl-2-NP) || || 0.447 (-0.155)<ref name="Schwarzenbach1990"/> || || || |
|- | |- | ||
| 2,4,6-trinitrotoluene (TNT) || -0.280 || || 2.869 || 5.204 || | | 2,4,6-trinitrotoluene (TNT) || -0.280 || || 2.869 || 5.204 || | ||
Revision as of 18:23, 4 April 2022
Abiotic Reduction of Munitions Constituents
Munition compounds (MCs) often contain one or more nitro (-NO2) functional groups which makes them susceptible to abiotic reduction, i.e., transformation by accepting electrons from a chemical electron donor. In soil and groundwater, the most prevalent electron donors are natural organic carbon and iron minerals. Understanding the kinetics and mechanisms of abiotic reduction of MCs by carbon and iron constituents in soil is not only essential for evaluating the environmental fate of MCs but also key to developing cost-efficient remediation strategies. This article summarizes the recent advances in our understanding of MC reduction by carbon and iron based reductants.
Related Article(s):
Contributor(s):
- Dr. Jimmy Murillo-Gelvez
- Paula Andrea Cárdenas-Hernández
- Dr. Pei Chiu
Key Resource(s):
- Schwarzenbach, Gschwend, and Imboden, 2016. Environmental Organic Chemistry, 3rd ed.[1]
Introduction
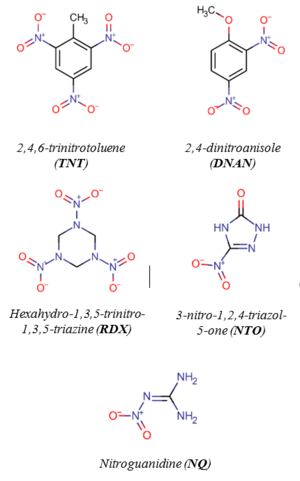
Legacy and insensitive MCs (Figure 1.) are susceptible to reductive transformation in soil and groundwater. Many redox-active constituents in the subsurface, especially those containing organic carbon, Fe(II), and sulfur can mediate MC reduction. Specific examples include Fe(II)-organic complexes[2][3][4][5][6], iron oxides in the presence of aqueous Fe(II)[7][8][9][10][11][12][13][14][15][16][17], magnetite[12][14][18][19][20], Fe(II)-bearing clays[21][22][23][24][25][26][27], hydroquinones (as surrogates of natural organic matter)[4][28][29][30][31][32][33], dissolved organic matter[34][35][36], black carbon[37][38][39][40][41][42], and sulfides[43][44]. These geo-reductants may control the fate and half-lives of MCs in the environment and can be used to promote MC degradation in soil and groundwater through enhanced natural attenuation[45].
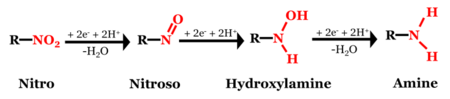
Although the chemical structures of MCs can vary significantly (Figure 1), most of them contain at least one nitro functional group (-NO2), which is susceptible to reductive transformation[46]. Of the MCs shown in Figure 1, 2,4,6-trinitrotoluene (TNT), 2,4-dinitroanisole (DNAN), and 3-nitro-1,2,4-triazol-5-one (NTO)[47] are nitroaromatic compounds (NACs) and hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) and nitroguanidine (NQ) are nitramines. The structural differences may result in different reactivities and reaction pathways. Reduction of NACs results in the formation of aromatic amines (i.e., anilines) with nitroso and hydroxylamine compounds as intermediates (Figure 2)[1].
Although the final reduction products are different for non-aromatic MCs, the reduction process often starts with the transformation of the -NO2 moiety, either through de-nitration (e.g., RDX[48][49]) or reduction to nitroso[33][50] followed by ring cleavage[6][49][50][51].
Figure 3 illustrates a typical MC reduction reaction. A redox-active soil constituent, such as organic matter or iron mineral, donates electrons to an MC and transforms the nitro group into an amino group (R-NH2). The rate at which an MC is reduced can vary by many orders of magnitude depending on the soil constituent, the MC, the reduction potential (EH) and other media conditions[52].
The most prevalent reductants in soils are iron minerals and organic carbon such as that found in natural organic matter. It has been suggested that Fe(II)aq and dissolved organic matter concentrations could serve as indicators of NAC reducibility in anaerobic sediments[53]. The following sections summarize these two classes of reductants separately and present advances in our understanding of the kinetics of NAC/MC reduction by these geo-reductants.
Carbonaceous Reductants
The two most predominant forms of organic carbon in natural systems are natural organic matter (NOM) and black carbon (BC)[54]. Black carbon includes charcoal, soot, graphite, and coal. Until the early 2000s black carbon was considered to be a class of (bio)chemically inert geosorbents[55]. However, it has been shown that BC can contain abundant quinone functional groups and thus can store and exchange electrons[56] with chemical[57] and biological[58] agents in the surroundings. Specifically, BC such as biochar has been shown to reductively transform MCs including NTO, DNAN, and RDX[42].
NOM encompasses all the organic compounds present in terrestrial and aquatic environments and can be classified into two groups, non-humic and humic substances. Humic substances (HS) contain a wide array of functional groups including carboxyl, enol, ether, ketone, ester, amide, (hydro)quinone, and phenol[59]. Quinone and hydroquinone groups are believed to be the predominant redox moieties responsible for the capacity of HS and BC to store and reversibly accept and donate electrons (i.e., through reduction and oxidation of HS/BC, respectively)[28][34][56][60][61][62][63][64][65][66][67][68][69][70][71].
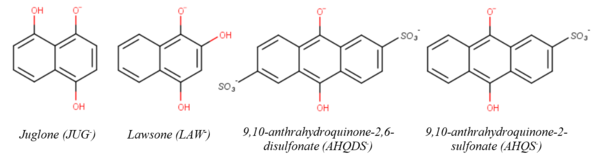
Hydroquinones have been widely used as surrogates to understand the reductive transformation of NACs and MCs by NOM. Figure 4 shows the chemical structures of the singly deprotonated forms of four hydroquinone species previously used to study NAC/MC reduction. The second-order rate constants (kR) for the reduction of NACs/MCs by these hydroquinone species are listed in Table 1, along with the aqueous-phase one electron reduction potentials of the NACs/MCs (EH1’) where available. EH1’ is an experimentally measurable thermodynamic property that reflects the propensity of a given NAC/MC to accept an electron in water (EH1(R-NO2)):
- Equation 1: R-NO2 + e- ⇔ R-NO2•-
Knowing the identity of and reaction order in the reductant is required to derive the second-order rate constants listed in Table 1. This same reason limits the utility of reduction rate constants measured with complex carbonaceous reductants such as NOM[34], BC[37][38][39][72], and HS[35][36], whose chemical structures and redox moieties responsible for the reduction, as well as their abundance, are not clearly defined or known. In other words, the observed rate constants in those studies are specific to the experimental conditions (e.g., pH and NOM source and concentration), and may not be easily comparable to other studies.
| Compound | EH1' (V) | Hydroquinone (log kR (M-1s-1)) | |||
|---|---|---|---|---|---|
| (NAC/MC) | LAW- | JUG- | AHQDS- | AHQS- | |
| Nitrobenzene (NB) | -0.485[28] | 0.380[28] | -1.102[28] | 2.050 | 3.060 |
| 2-nitrotoluene (2-NT) | -0.590[28] | -1.432[28] | -2.523[28] | 0.775 | |
| 3-nitrotoluene (3-NT) | -0.475[28] | 0.462[28] | -0.921[28] | ||
| 4-nitrotoluene (4-NT) | -0.500[28] | 0.041[28] | -1.292[28] | 1.822 | 2.610 |
| 2-chloronitrobenzene (2-ClNB) | -0.485[28] | 0.342[28] | -0.824[28] | 2.412 | |
| 3-chloronitrobenzene (3-ClNB) | -0.405[28] | 1.491[28] | 0.114[28] | ||
| 4-chloronitrobenzene (4-ClNB) | -0.450[28] | 1.041[28] | -0.301[28] | 2.988 | |
| 2-acetylnitrobenzene (2-AcNB) | -0.470[28] | 0.519[28] | -0.456[28] | ||
| 3-acetylnitrobenzene (3-AcNB) | -0.405[28] | 1.663[28] | 0.398[28] | ||
| 4-acetylnitrobenzene (4-AcNB) | -0.360[28] | 2.519[28] | 1.477[28] | ||
| 2-nitrophenol (2-NP) | 0.568 (0.079)[28] | ||||
| 4-nitrophenol (4-NP) | -0.699 (-1.301)[28] | ||||
| 4-methyl-2-nitrophenol (4-Me-2-NP) | 0.748 (0.176)[28] | ||||
| 4-chloro-2-nitrophenol (4-Cl-2-NP) | 1.602 (1.114)[28] | ||||
| 5-fluoro-2-nitrophenol (5-Cl-2-NP) | 0.447 (-0.155)[28] | ||||
| 2,4,6-trinitrotoluene (TNT) | -0.280 | 2.869 | 5.204 | ||
| 2-amino-4,6-dinitrotoluene (2-A-4,6-DNT) | -0.400 | 0.987 | |||
| 4-amino-2,6-dinitrotoluene (4-A-2,6-DNT) | -0.440 | 0.079 | |||
| 2,4-diamino-6-nitrotoluene (2,4-DA-6-NT) | -0.505 | -1.678 | |||
| 2,6-diamino-4-nitrotoluene (2,6-DA-4-NT) | -0.495 | -1.252 | |||
| 1,3-dinitrobenzene (1,3-DNB) | -0.345 | 1.785 | |||
| 1,4-dinitrobenzene (1,4-DNB) | -0.257 | 3.839 | |||
| 2-nitroaniline (2-NANE) | < -0.560 | -2.638 | |||
| 3-nitroaniline (3-NANE) | -0.500 | -1.367 | |||
| 1,2-dinitrobenzene (1,2-DNB) | -0.290 | 5.407 | |||
| 4-nitroanisole (4-NAN) | -0.661 | 1.220 | |||
| 2-amino-4-nitroanisole (2-A-4-NAN) | -0.924 | 1.150 | 2.190 | ||
| 4-amino-2-nitroanisole (4-A-2-NAN) | 1.610 | 2.360 | |||
| 2-chloro-4-nitroaniline (2-Cl-5-NANE) | -0.863 | 1.250 | 2.210 | ||
| N-methyl-4-nitroaniline (MNA) | -1.740 | -0.260 | 0.692 | ||
| 3-nitro-1,2,4-triazol-5-one (NTO) | 5.701 (1.914) | ||||
| Hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) | -0.349 | ||||
Advantages and Limitations of the Technology for PFAS Treatment
Advantages:
- High removal rates of long-chain PFAS (C5-C8) due to the production of versatile reactive species
- Requires no chemical additions and produces no residual waste
- Total organic carbon (TOC) concentration and other non-surfactant co-contaminants do not influence the process efficiency
- The process is mobile and scalable
- Versatile: can be used in batch and continuous systems
Limitations:
- Limited removal of short-chain PFAS due to their inability to concentrate at plasma-liquid interfaces. Addition of surfactants such as CTAB improves their removal and degradation rates.
- Excessive foaming caused by bubbling argon gas through a solution containing high (>10 mg/L) concentrations of long-chain (surfactant) PFAS may interfere with the formation of plasma.
Summary
PFAS are susceptible to plasma treatment because the hydrophobic PFAS accumulates at the gas-liquid interface, exposing more of the PFAS to the plasma. Plasma-based treatment of PFAS contaminated water successfully degrades PFOA and PFOS to below the EPA health advisory level of 70 ppt and accomplishes the near complete destruction of other PFAS within a short treatment time. PFAS concentration reductions of ≥90% and post-treatment concentrations below laboratory detection levels are common for long chain PFAS and precursors. The lack of sensitivity of plasma to co-contaminants, coupled with high PFAS removal and defluorination efficiencies, makes plasma-based water treatment a promising technology for the remediation of PFAS-contaminated water. The plasma treatment process is currently developed for ex situ application and can also be integrated into a treatment train[73].
References
- ^ 1.0 1.1 Schwarzenbach, R.P., Gschwend, P.M., and Imboden, D.M., 2016. Environmental Organic Chemistry, 3rd Edition. John Wiley and Sons, Ltd, 1024 pages. ISBN: 978-1-118-76723-8
- ^ Naka, D., Kim, D., and Strathmann, T.J., 2006. Abiotic Reduction of Nitroaromatic Compounds by Aqueous Iron(II)−Catechol Complexes. Environmental Science and Technology 40(9), pp. 3006–3012. DOI: 10.1021/es060044t
- ^ Naka, D., Kim, D., Carbonaro, R.F., and Strathmann, T.J., 2008. Abiotic reduction of nitroaromatic contaminants by iron(II) complexes with organothiol ligands. Environmental Toxicology and Chemistry, 27(6), pp. 1257–1266. DOI: 10.1897/07-505.1
- ^ 4.0 4.1 Hartenbach, A.E., Hofstetter, T.B., Aeschbacher, M., Sander, M., Kim, D., Strathmann, T.J., Arnold, W.A., Cramer, C.J., and Schwarzenbach, R.P., 2008. Variability of Nitrogen Isotope Fractionation during the Reduction of Nitroaromatic Compounds with Dissolved Reductants. Environmental Science and Technology 42(22), pp. 8352–8359. DOI: 10.1021/es801063u
- ^ Kim, D., Duckworth, O.W., and Strathmann, T.J., 2009. Hydroxamate siderophore-promoted reactions between iron(II) and nitroaromatic groundwater contaminants. Geochimica et Cosmochimica Acta, 73(5), pp. 1297–1311. DOI: 10.1016/j.gca.2008.11.039
- ^ 6.0 6.1 Kim, D., and Strathmann, T.J., 2007. Role of Organically Complexed Iron(II) Species in the Reductive Transformation of RDX in Anoxic Environments. Environmental Science and Technology, 41(4), pp. 1257–1264. DOI: 10.1021/es062365a
- ^ Colón, D., Weber, E.J., and Anderson, J.L., 2006. QSAR Study of the Reduction of Nitroaromatics by Fe(II) Species. Environmental Science and Technology, 40(16), pp. 4976–4982. DOI: 10.1021/es052425x
- ^ Luan, F., Xie, L., Li, J., and Zhou, Q., 2013. Abiotic reduction of nitroaromatic compounds by Fe(II) associated with iron oxides and humic acid. Chemosphere, 91(7), pp. 1035–1041. DOI: 10.1016/j.chemosphere.2013.01.070
- ^ Gorski, C.A., Edwards, R., Sander, M., Hofstetter, T.B., and Stewart, S.M., 2016. Thermodynamic Characterization of Iron Oxide–Aqueous Fe2+ Redox Couples. Environmental Science and Technology, 50(16), pp. 8538–8547. DOI: 10.1021/acs.est.6b02661
- ^ Fan, D., Bradley, M.J., Hinkle, A.W., Johnson, R.L., and Tratnyek, P.G., 2016. Chemical Reactivity Probes for Assessing Abiotic Natural Attenuation by Reducing Iron Minerals. Environmental Science and Technology, 50(4), pp. 1868–1876. DOI: 10.1021/acs.est.5b05800
- ^ Jones, A.M., Kinsela, A.S., Collins, R.N., and Waite, T.D., 2016. The reduction of 4-chloronitrobenzene by Fe(II)-Fe(III) oxide systems - correlations with reduction potential and inhibition by silicate. Journal of Hazardous Materials, 320, pp. 143–149. DOI: 10.1016/j.jhazmat.2016.08.031
- ^ 12.0 12.1 Klausen, J., Troeber, S.P., Haderlein, S.B., and Schwarzenbach, R.P., 1995. Reduction of Substituted Nitrobenzenes by Fe(II) in Aqueous Mineral Suspensions. Environmental Science and Technology, 29(9), pp. 2396–2404. DOI: 10.1021/es00009a036
- ^ Strehlau, J.H., Stemig, M.S., Penn, R.L., and Arnold, W.A., 2016. Facet-Dependent Oxidative Goethite Growth As a Function of Aqueous Solution Conditions. Environmental Science and Technology, 50(19), pp. 10406–10412. DOI: 10.1021/acs.est.6b02436
- ^ 14.0 14.1 Elsner, M., Schwarzenbach, R.P., and Haderlein, S.B., 2004. Reactivity of Fe(II)-Bearing Minerals toward Reductive Transformation of Organic Contaminants. Environmental Science and Technology, 38(3), pp. 799–807. DOI: 10.1021/es0345569
- ^ Colón, D., Weber, E.J., and Anderson, J.L., 2008. Effect of Natural Organic Matter on the Reduction of Nitroaromatics by Fe(II) Species. Environmental Science and Technology, 42(17), pp. 6538–6543. DOI: 10.1021/es8004249
- ^ Stewart, S.M., Hofstetter, T.B., Joshi, P. and Gorski, C.A., 2018. Linking Thermodynamics to Pollutant Reduction Kinetics by Fe2+ Bound to Iron Oxides. Environmental Science and Technology, 52(10), pp. 5600–5609. DOI: 10.1021/acs.est.8b00481 Open access article.
- ^ Klupinski, T.P., Chin, Y.P., and Traina, S.J., 2004. Abiotic Degradation of Pentachloronitrobenzene by Fe(II): Reactions on Goethite and Iron Oxide Nanoparticles. Environmental Science and Technology, 38(16), pp. 4353–4360. DOI: 10.1021/es035434j
- ^ Heijman, C.G., Holliger, C., Glaus, M.A., Schwarzenbach, R.P., and Zeyer, J., 1993. Abiotic Reduction of 4-Chloronitrobenzene to 4-Chloroaniline in a Dissimilatory Iron-Reducing Enrichment Culture. Applied and Environmental Microbiology, 59(12), pp. 4350–4353. DOI: 10.1128/aem.59.12.4350-4353.1993 Open access article.
- ^ Gorski, C.A., and Scherer, M.M., 2009. Influence of Magnetite Stoichiometry on FeII Uptake and Nitrobenzene Reduction. Environmental Science and Technology, 43(10), pp. 3675–3680. DOI: 10.1021/es803613a
- ^ Gorski, C.A., Nurmi, J.T., Tratnyek, P.G., Hofstetter, T.B. and Scherer, M.M., 2010. Redox Behavior of Magnetite: Implications for Contaminant Reduction. Environmental Science and Technology, 44(1), pp. 55–60. DOI: 10.1021/es9016848
- ^ Hofstetter, T.B., Neumann, A., and Schwarzenbach, R.P., 2006. Reduction of Nitroaromatic Compounds by Fe(II) Species Associated with Iron-Rich Smectites. Environmental Science and Technology, 40(1), pp. 235–242. DOI: 10.1021/es0515147
- ^ Schultz, C. A., and Grundl, T.J., 2000. pH Dependence on Reduction Rate of 4-Cl-Nitrobenzene by Fe(II)/Montmorillonite Systems. Environmental Science and Technology 34(17), pp. 3641–3648. DOI: 10.1021/es990931e
- ^ Luan, F., Gorski, C.A., and Burgos, W.D., 2015. Linear Free Energy Relationships for the Biotic and Abiotic Reduction of Nitroaromatic Compounds. Environmental Science and Technology, 49(6), pp. 3557–3565. DOI: 10.1021/es5060918
- ^ Luan, F., Liu, Y., Griffin, A.M., Gorski, C.A. and Burgos, W.D., 2015. Iron(III)-Bearing Clay Minerals Enhance Bioreduction of Nitrobenzene by Shewanella putrefaciens CN32. Environmental Science and Technology, 49(3), pp. 1418–1426. DOI: 10.1021/es504149y
- ^ Hofstetter, T.B., Schwarzenbach, R.P. and Haderlein, S.B., 2003. Reactivity of Fe(II) Species Associated with Clay Minerals. Environmental Science and Technology, 37(3), pp. 519–528. DOI: 10.1021/es025955r
- ^ Neumann, A., Hofstetter, T.B., Lüssi, M., Cirpka, O.A., Petit, S., and Schwarzenbach, R.P., 2008. Assessing the Redox Reactivity of Structural Iron in Smectites Using Nitroaromatic Compounds As Kinetic Probes. Environmental Science and Technology, 42(22), pp. 8381–8387. DOI: 10.1021/es801840x
- ^ Hofstetter, T.B., Neumann, A., Arnold, W.A., Hartenbach, A.E., Bolotin, J., Cramer, C.J., and Schwarzenbach, R.P., 2008. Substituent Effects on Nitrogen Isotope Fractionation During Abiotic Reduction of Nitroaromatic Compounds. Environmental Science and Technology, 42(6), pp. 1997–2003. DOI: 10.1021/es702471k
- ^ 28.00 28.01 28.02 28.03 28.04 28.05 28.06 28.07 28.08 28.09 28.10 28.11 28.12 28.13 28.14 28.15 28.16 28.17 28.18 28.19 28.20 28.21 28.22 28.23 28.24 28.25 28.26 28.27 28.28 28.29 28.30 28.31 28.32 28.33 28.34 28.35 28.36 Schwarzenbach, R.P., Stierli, R., Lanz, K., and Zeyer, J., 1990. Quinone and Iron Porphyrin Mediated Reduction of Nitroaromatic Compounds in Homogeneous Aqueous Solution. Environmental Science and Technology, 24(10), pp. 1566–1574. DOI: 10.1021/es00080a017
- ^ Tratnyek, P.G., and Macalady, D.L., 1989. Abiotic Reduction of Nitro Aromatic Pesticides in Anaerobic Laboratory Systems. Journal of Agricultural and Food Chemistry, 37(1), pp. 248–254. DOI: 10.1021/jf00085a058
- ^ Hofstetter, T.B., Heijman, C.G., Haderlein, S.B., Holliger, C. and Schwarzenbach, R.P., 1999. Complete Reduction of TNT and Other (Poly)nitroaromatic Compounds under Iron-Reducing Subsurface Conditions. Environmental Science and Technology, 33(9), pp. 1479–1487. DOI: 10.1021/es9809760
- ^ Murillo-Gelvez, J., Hickey, K.P., Di Toro, D.M., Allen, H.E., Carbonaro, R.F., and Chiu, P.C., 2019. Experimental Validation of Hydrogen Atom Transfer Gibbs Free Energy as a Predictor of Nitroaromatic Reduction Rate Constants. Environmental Science and Technology, 53(10), pp. 5816–5827. DOI: 10.1021/acs.est.9b00910
- ^ Niedźwiecka, J.B., Drew, S.R., Schlautman, M.A., Millerick, K.A., Grubbs, E., Tharayil, N. and Finneran, K.T., 2017. Iron and Electron Shuttle Mediated (Bio)degradation of 2,4-Dinitroanisole (DNAN). Environmental Science and Technology, 51(18), pp. 10729–10735. DOI: 10.1021/acs.est.7b02433
- ^ 33.0 33.1 Kwon, M.J., and Finneran, K.T., 2006. Microbially Mediated Biodegradation of Hexahydro-1,3,5-Trinitro-1,3,5- Triazine by Extracellular Electron Shuttling Compounds. Applied and Environmental Microbiology, 72(9), pp. 5933–5941. DOI: 10.1128/AEM.00660-06 Open access article.
- ^ 34.0 34.1 34.2 Dunnivant, F.M., Schwarzenbach, R.P., and Macalady, D.L., 1992. Reduction of Substituted Nitrobenzenes in Aqueous Solutions Containing Natural Organic Matter. Environmental Science and Technology, 26(11), pp. 2133–2141. DOI: 10.1021/es00035a010
- ^ 35.0 35.1 Luan, F., Burgos, W.D., Xie, L., and Zhou, Q., 2010. Bioreduction of Nitrobenzene, Natural Organic Matter, and Hematite by Shewanella putrefaciens CN32. Environmental Science and Technology, 44(1), pp. 184–190. DOI: 10.1021/es901585z
- ^ 36.0 36.1 Murillo-Gelvez, J., di Toro, D.M., Allen, H.E., Carbonaro, R.F., and Chiu, P.C., 2021. Reductive Transformation of 3-Nitro-1,2,4-triazol-5-one (NTO) by Leonardite Humic Acid and Anthraquinone-2,6-disulfonate (AQDS). Environmental Science and Technology, 55(19), pp. 12973–12983. DOI: 10.1021/acs.est.1c03333
- ^ 37.0 37.1 Oh, S.-Y., Son, J.G., and Chiu, P.C., 2013. Biochar-Mediated Reductive Transformation of Nitro Herbicides and Explosives. Environmental Toxicology and Chemistry, 32(3), pp. 501–508. DOI: 10.1002/etc.2087 Open access article.
- ^ 38.0 38.1 Oh, S.-Y., and Chiu, P.C., 2009. Graphite- and Soot-Mediated Reduction of 2,4-Dinitrotoluene and Hexahydro-1,3,5-trinitro-1,3,5-triazine. Environmental Science & Technology, 43(18), pp. 6983–6988. DOI: 10.1021/es901433m
- ^ 39.0 39.1 Xu, W., Pignatello, J.J., and Mitch, W.A., 2015. Reduction of Nitroaromatics Sorbed to Black Carbon by Direct Reaction with Sorbed Sulfides. Environmental Science and Technology, 49(6), pp. 3419–3426. DOI: 10.1021/es5045198
- ^ Oh, S.-Y., Cha, D.K., and Chiu, P.C., 2002. Graphite-Mediated Reduction of 2,4-Dinitrotoluene with Elemental Iron. Environmental Science and Technology, 36(10), pp. 2178–2184. DOI: 10.1021/es011474g
- ^ Amezquita-Garcia, H.J., Razo-Flores, E., Cervantes, F.J., and Rangel-Mendez, J.R., 2013. Activated carbon fibers as redox mediators for the increased reduction of nitroaromatics. Carbon, 55, pp. 276–284. DOI: 10.1016/j.carbon.2012.12.062
- ^ 42.0 42.1 Xin, D., Girón, J., Fuller, M.E., and Chiu, P.C., 2022. Abiotic Reduction of 3-Nitro-1,2,4-triazol-5-one (NTO) and Other Munitions Constituents by Wood-Derived Biochar through Its Rechargeable Electron Storage Capacity. Environmental Science: Processes and Impacts, 24(2), pp. 316-329. DOI: 10.1039/D1EM00447F
- ^ Hojo, M., Takagi, Y. and Ogata, Y., 1960. Kinetics of the Reduction of Nitrobenzenes by Sodium Disulfide. Journal of the American Chemical Society, 82(10), pp. 2459–2462. DOI: 10.1021/ja01495a017
- ^ Zeng, T., Chin, Y.P., and Arnold, W.A., 2012. Potential for Abiotic Reduction of Pesticides in Prairie Pothole Porewaters. Environmental Science and Technology, 46(6), pp. 3177–3187. DOI: 10.1021/es203584d
- ^ US EPA, 2012. A Citizen’s Guide to Monitored Natural Attenuation. EPA document 542-F-12-014. Free download.
- ^ Spain, J.C., Hughes, J.B., and Knackmuss, H.J., 2000. Biodegradation of Nitroaromatic Compounds and Explosives. CRC Press, 456 pages. ISBN: 9780367398491
- ^ Harris, N.J., and Lammertsma, K., 1996. Tautomerism, Ionization, and Bond Dissociations of 5-Nitro-2,4-dihydro-3H-1,2,4-triazolone. Journal of the American Chemical Society, 118(34), pp. 8048–8055. DOI: 10.1021/ja960834a
- ^ Kwon, M.J., and Finneran, K.T., 2008. Biotransformation products and mineralization potential for hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) in abiotic versus biological degradation pathways with anthraquinone-2,6-disulfonate (AQDS) and Geobacter metallireducens. Biodegradation, 19(5), pp. 705–715. DOI: 10.1007/s10532-008-9175-5
- ^ 49.0 49.1 Halasz, A., and Hawari, J., 2011. Degradation Routes of RDX in Various Redox Systems. Aquatic Redox Chemistry, American Chemical Society, 1071(20), pp. 441-462. DOI: 10.1021/bk-2011-1071.ch020
- ^ 50.0 50.1 Tong, Y., Berens, M.J., Ulrich, B.A., Bolotin, J., Strehlau, J.H., Hofstetter, T.B., and Arnold, W.A., 2021. Exploring the Utility of Compound-Specific Isotope Analysis for Assessing Ferrous Iron-Mediated Reduction of RDX in the Subsurface. Environmental Science and Technology, 55(10), pp. 6752–6763. DOI: 10.1021/acs.est.0c08420
- ^ Larese-Casanova, P., and Scherer, M.M., 2008. Abiotic Transformation of Hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) by Green Rusts. Environmental Science and Technology, 42(11), pp. 3975–3981. DOI: 10.1021/es702390b
- ^ Borch, T., Kretzschmar, R., Kappler, A., Cappellen, P.V., Ginder-Vogel, M., Voegelin, A., and Campbell, K., 2010. Biogeochemical Redox Processes and their Impact on Contaminant Dynamics. Environmental Science and Technology, 44(1), pp. 15–23. DOI: 10.1021/es9026248 Open access article.
- ^ Zhang, H., and Weber, E.J., 2009. Elucidating the Role of Electron Shuttles in Reductive Transformations in Anaerobic Sediments. Environmental Science and Technology, 43(4), pp. 1042–1048. DOI: 10.1021/es8017072
- ^ Schumacher, B.A., 2002. Methods for the Determination of Total Organic Carbon (TOC) in Soils and Sediments. U.S. EPA, Ecological Risk Assessment Support Center. Free download.
- ^ Schmidt, M.W.I., and Noack, A.G., 2000. Black carbon in soils and sediments: Analysis, distribution, implications, and current challenges. Global Biogeochemical Cycles, 14(3), pp. 777–793. DOI: 10.1029/1999GB001208 Open access article.
- ^ 56.0 56.1 Klüpfel, L., Keiluweit, M., Kleber, M., and Sander, M., 2014. Redox Properties of Plant Biomass-Derived Black Carbon (Biochar). Environmental Science and Technology, 48(10), pp. 5601–5611. DOI: 10.1021/es500906d
- ^ Xin, D., Xian, M., and Chiu, P.C., 2019. New methods for assessing electron storage capacity and redox reversibility of biochar. Chemosphere, 215, 827–834. DOI: 10.1016/j.chemosphere.2018.10.080
- ^ Saquing, J.M., Yu, Y.-H., and Chiu, P.C., 2016. Wood-Derived Black Carbon (Biochar) as a Microbial Electron Donor and Acceptor. Environmental Science and Technology Letters, 3(2), pp. 62–66. DOI: 10.1021/acs.estlett.5b00354
- ^ Sparks, D.L., 2003. Environmental Soil Chemistry, 2nd Edition. Elsevier Science and Technology Books. DOI: 10.1016/B978-0-12-656446-4.X5000-2
- ^ Scott, D.T., McKnight, D.M., Blunt-Harris, E.L., Kolesar, S.E., and Lovley, D.R., 1998. Quinone Moieties Act as Electron Acceptors in the Reduction of Humic Substances by Humics-Reducing Microorganisms. Environmental Science and Technology, 32(19), pp. 2984–2989. DOI: 10.1021/es980272q
- ^ Cory, R.M., and McKnight, D.M., 2005. Fluorescence Spectroscopy Reveals Ubiquitous Presence of Oxidized and Reduced Quinones in Dissolved Organic Matter. Environmental Science & Technology, 39(21), pp 8142–8149. DOI: 10.1021/es0506962
- ^ Fimmen, R.L., Cory, R.M., Chin, Y.P., Trouts, T.D., and McKnight, D.M., 2007. Probing the oxidation–reduction properties of terrestrially and microbially derived dissolved organic matter. Geochimica et Cosmochimica Acta, 71(12), pp. 3003–3015. DOI: 10.1016/j.gca.2007.04.009
- ^ Struyk, Z., and Sposito, G., 2001. Redox properties of standard humic acids. Geoderma, 102(3-4), pp. 329–346. DOI: 10.1016/S0016-7061(01)00040-4
- ^ Ratasuk, N., and Nanny, M.A., 2007. Characterization and Quantification of Reversible Redox Sites in Humic Substances. Environmental Science and Technology, 41(22), pp. 7844–7850. DOI: 10.1021/es071389u
- ^ Aeschbacher, M., Sander, M., and Schwarzenbach, R.P., 2010. Novel Electrochemical Approach to Assess the Redox Properties of Humic Substances. Environmental Science and Technology, 44(1), pp. 87–93. DOI: 10.1021/es902627p
- ^ Aeschbacher, M., Vergari, D., Schwarzenbach, R.P., and Sander, M., 2011. Electrochemical Analysis of Proton and Electron Transfer Equilibria of the Reducible Moieties in Humic Acids. Environmental Science and Technology, 45(19), pp. 8385–8394. DOI: 10.1021/es201981g
- ^ Bauer, I., and Kappler, A., 2009. Rates and Extent of Reduction of Fe(III) Compounds and O2 by Humic Substances. Environmental Science and Technology, 43(13), pp. 4902–4908. DOI: 10.1021/es900179s
- ^ Maurer, F., Christl, I. and Kretzschmar, R., 2010. Reduction and Reoxidation of Humic Acid: Influence on Spectroscopic Properties and Proton Binding. Environmental Science and Technology, 44(15), pp. 5787–5792. DOI: 10.1021/es100594t
- ^ Walpen, N., Schroth, M.H., and Sander, M., 2016. Quantification of Phenolic Antioxidant Moieties in Dissolved Organic Matter by Flow-Injection Analysis with Electrochemical Detection. Environmental Science and Technology, 50(12), pp. 6423–6432. DOI: 10.1021/acs.est.6b01120 Open access article.
- ^ Aeschbacher, M., Graf, C., Schwarzenbach, R.P., and Sander, M., 2012. Antioxidant Properties of Humic Substances. Environmental Science and Technology, 46(9), pp. 4916–4925. DOI: 10.1021/es300039h
- ^ Nurmi, J.T., and Tratnyek, P.G., 2002. Electrochemical Properties of Natural Organic Matter (NOM), Fractions of NOM, and Model Biogeochemical Electron Shuttles. Environmental Science and Technology, 36(4), pp. 617–624. DOI: 10.1021/es0110731
- ^ Xin, D., 2021. Understanding the Electron Storage Capacity of Pyrogenic Black Carbon: Origin, Redox Reversibility, Spatial Distribution, and Environmental Applications. Doctoral Thesis, University of Delaware. Free download.
- ^ Richardson, S., 2021. Nanofiltration Followed by Electrical Discharge Plasma for Destruction of PFAS and Co-occurring Chemicals in Groundwater: A Treatment Train Approach. Environmental Security Technology Certification Program (ESTCP), Project Number ER21-5136. Project Overview