Difference between revisions of "User:Debra Tabron/sandbox"
Debra Tabron (talk | contribs) |
Debra Tabron (talk | contribs) |
||
| Line 44: | Line 44: | ||
|- | |- | ||
|} | |} | ||
| + | ==Behavior in Groundwater== | ||
| + | Dissolved metal behavior in groundwater is controlled by reactions within the aqueous phase and reactions between the contaminant metal and the solid phase (such as clay particles or sand grains in the aquifer). Hence, the chemical composition of the water and the aquifer mineralogy are important to metal contaminant behavior. In turn, the water chemistry can be controlled by the aquifer mineralogy, the chemical composition of the infiltrating plume, or reactions between the infiltrating plume and aquifer minerals. If carbon and other nutrients are available, reactions controlling metal behavior can be influenced by microbial activity. | ||
| + | All metal contaminants in groundwater are distributed among dissolved species in which the metal ion (an ion is a dissolved species with a positive or negative charge) is associated with one or more ions, producing a different dissolved species with different chemical behavior<ref name="Langmuir1997">Langmuir, D., 1997, Aqueous Environmental Geochemistry. Prentice-Hall, Inc. Upper Saddle River, NJ ISBN: 978-0023674129.</ref><ref>Drever, J.I., The Geochemistry of Natural Waters: Surface and Groundwater Environments. Prentice-Hall, Inc., ISBN 0132727900.</ref>. For example, the mercury ion, Hg<sup>+2</sup>, readily associates with the chloride ion, Cl<sup>-</sup>, to produce a series of species (HgCl<sup>+</sup>, HgCl2°, HgCl<sub>3</sub><sup>-</sup>) with concentrations dependent on the chemical composition of the groundwater. The collection of species is called the aqueous [https://en.wikipedia.org/wiki/Speciation '''speciation'''] of the contaminant metal. Some contaminants have a simple aqueous speciation and few species dominate over a broad range of groundwater conditions. Others have a complicated aqueous speciation that changes with pH, redox potential, and/or the presence or absence of certain ions. Uranium is an example of a complicated aqueous speciation in common aquifer conditions (Fig. 1). The aqueous speciation strongly influences sorption and precipitation reactions involving the contaminant metal, and thus influences the mobility of the metal in the aquifer and the methods of effective remediation (see article on '''Mobility of Metals and Metalloids'''). | ||
| + | [[File:Denham-Article 1-Figure 1. PNG.PNG|500px|thumbnail|right|Figure 1: Uranium’s complicated aqueous speciation (diagram produced with The Geochemist’s Workbench®<ref>Bethke, C.M. and S. Yeakel, 2015. The geochemist’s workbench®, Release 10.0. Latest version available at [www.gwb.com www.gwb.com]</ref>, PCO<sub>2</sub> = 0.01 atm., [Ca] = 10 (mg/L).]] | ||
| + | The most important aspect of a metal contaminants aqueous speciation is whether multiple oxidation states can occur over the range of conditions found in groundwater. Contaminants that have multiple oxidation states that are stable in groundwater are considered “redox sensitive”. Different oxidation states of a metal can have very different behaviors and toxicity in groundwater. For example, chromium in a plus six oxidation state (chromate) is more mobile and toxic than chromium in a plus three oxidation state. Table 2 lists whether several common metal contaminants are redox sensitive and the relative complexity of their aqueous speciation. Here, a complicated aqueous speciation is defined as having 4 or more species that dominate various portions of the field defined by a pH range of 4 to 10 and an Eh (Eh is a measure of the tendency of electrons to flow from one ion to another<ref name = "Hem1970">Hem, J.D., 1970. Chemical behavior of mercury in aqueous media. In Mercury in the Environment (Vol. 713, pp. 19-24). Washington, DC: US Government Printing Office. [http://www.environmentalrestoration.wiki/images/8/82/Hem-1970-Chemical_behavior_of_mercury_in_aqueous_media.pdf Report pdf]</ref> that covers the stability field of water (as determined using the thermodynamic database “thermo_minteq” and The Geochemist’s Workbench® with [Cl<sup>-</sup>] and [SO<sub>4</sub><sup>-2</sup>] = 10 mg/L). The complexity of aqueous speciation is not determined by redox sensitivity because many metals that do not change oxidation states with varying redox conditions may still form aqueous sulfide or bisulfide species under sufficiently reducing conditions. | ||
| + | The importance of aqueous speciation of a contaminant metal to environmental remediation can be illustrated by the behavior of uranium. Many groundwaters contaminated by uranium contain dissolved oxygen and moderate concentrations of dissolved carbon dioxide. Under these conditions, the dominant aqueous species of oxidized uranium [U(VI)] are neutrally or negatively charged carbonate species (Fig. 1). These species tend to keep oxidized uranium in the dissolved state, making it difficult to limit its mobility by causing it to adsorb or precipitate in a solid phase. The challenge is to find some method that either overcomes the effects of the carbonate species or changes the speciation of the uranium. | ||
| + | One method that has been studied extensively is to change the speciation by converting the uranium to the reduced form, U(IV), causing the uranium to precipitate as a low solubility U(IV) oxide. This can be done with chemicals (abiotic reduction) or by stimulating indigenous microbes to consume all of the oxygen in the groundwater (bioreduction). A common problem with this approach is that the groundwater naturally contains oxygen, and oxygenated groundwater will quickly flow into the treated zone re-oxidizing and remobilizing the uranium. | ||
| + | |||
| + | At some sites, oxidized uranium has been successfully treated with phosphate minerals because, under the conditions of the site groundwater, U(VI) is bound more strongly in the phosphate minerals than it is in the aqueous carbonate complexes. There are other ways to overcome the effects of the carbonate complexes and limit the mobility of uranium. The key idea to selecting a uranium remediation method that is likely to be successful, is understanding how the aqueous speciation of uranium will affect the treatment. | ||
Revision as of 15:23, 1 November 2016
Metals and metalloids (an element such as arsenic or antimony with properties in between those of metals and nonmetals are common groundwater contaminants that present a risk to users of groundwater if concentrations exceed acceptable risk-based concentrations. Contamination of groundwater by metals and metalloids is most often related to industrial sources or mining and mineral processing. Their acute and chronic toxicity as well as their common occurrence, make metal and metalloid contamination environmentally significant. The behavior and toxicity of metals and metalloids can vary with the chemical composition of the groundwater and the minerals present in the aquifer. Under some conditions, a given metal or metalloid may pose little risk because it is adsorbed to the aquifer solids or precipitated from groundwater before it reaches any exposure point. In other cases these contaminants may be quite mobile, forming groundwater plumes that can pose a risk to groundwater users.
Related Articles:
- Mobility of Metals and Metalloid Contaminants in Groundwater
- Monitored Natural Attenuation (MNA) of Metals and Metalloids
- Remediation of Metals and Metalloids
CONTRIBUTOR(S): Dr. Miles Denham
Key Resource(s):
Introduction
Groundwater contaminated with metals and metalloids (collectively referred to as “metals” in this article for simplicity; Table 1) can be a major problem because of the broad spectrum of sources, the toxicity of many metals, and the difficulty in remediating metal-contaminated sites. Common metal contaminants, their primary sources, and their potential health effects, especially those that occur at U.S. Department of Defense[1][2][3], U.S. Department of Energy[4][5], and private sites[6][7] are quite variable (e.g., Table 1). The behavior of metal contaminants in groundwater depends on the nature of the source, the chemistry of the metal, and the mineralogy of the aquifer. Regulation and remediation of metal contaminants is further complicated by the fact that metals occur naturally in aquifers, sometimes at levels that are safe for ingestion, but at other times at concentrations that naturally exceed acceptable risk-based concentrations such as Maximum Concentration Levels (MCLs).
| Metal or Metalloid* | Potential Natural and Man-Made Sources to Groundwater | Potential Health and Other Effects If Concentrations are Above Risk-Based Levels | MCL[8](mg/L) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Antimony* | Enters environment from natural weathering, industrial production, municipal waste disposal, and manufacturing of flame retardants, ceramics, glass, batteries, fireworks, explosives, and ammunition. | Decreases longevity, alters blood levels of glucose and cholesterol in laboratory animals exposed at high levels over their lifetime. | 0.006 | ||||||||
| Arsenic* | Enters environment from natural processes, industrial activities, pesticides, industrial waste, smelting of copper, lead, and zinc. | Causes acute and chronic toxicity, liver and kidney damage, decreases hemoglobin, carcinogenic. | 0.010 | ||||||||
| Cadmium | May enter the environment from industrial discharge, mining waste, metal/metalloid plating, water pipes, batteries, paints and pigments, plastic stabilizers, and landfill leachate. | Replaces zinc biochemically in the body and causes high blood pressure, liver and kidney damage. Destroys testicular tissue and red blood cells. Toxic to aquatic biota. | 0.005 | ||||||||
| Chromium | Used in metal/metalloid plating and as a cooling water additive. Also may enter environment from old mining operations runoff and leaching into groundwater. | Chromium (III) is a nutritionally essential element. Chromium (VI) is more toxic, causing liver and kidney damage, internal hemorrhaging, respiratory damage, dermatitis, and ulcers on the skin at high concentrations. | 0.1 (total) | ||||||||
| Copper | Enters environment from metal/metalloid plating, industrial and domestic waste, mining, and mineral leaching. | An essential nutrient in low doses. High doses can cause stomach and intestinal distress, liver and kidney damage, anemia. Essential trace element but toxic to plants and algae at moderate doses. | 1.3 | ||||||||
| Lead | Enters environment from industry, mining, plumbing, leaded-gasoline, and recycling of lead-acid batteries. | Affects red blood cell chemistry; delays normal physical and mental development in babies and young children. Can cause deficits in attention span, hearing, and learning in children. | 0.015 | ||||||||
| Mercury | Enters environment from industrial waste (e.g., chloralkali process), mining, pesticides, coal, electrical equipment e.g., batteries, lamps, switches), smelting, and fossil-fuel combustion. | Causes acute and chronic toxicity. Targets the kidneys and can cause nervous system disorders. | 0.002 | ||||||||
| Uranium | Enters groundwater from uranium mining and ore processing operations, nuclear fuel reprocessing facilities, and potentially from exploded depleted uranium munitions. | Depresses renal function, can cause kidney damage and failure in extreme cases. More of a toxicological problem than a radiation problem, though long-term exposure can increase risk of cancer. | 0.030 | ||||||||
| Zinc | Enters environment from industrial waste, metal/metalloid plating, plumbing, and is a major component of various industrial sludges. | Essential nutrient. Causes detrimental effects in humans at high doses. Can be toxic to aquatic organisms. | 5[9](secondary) | ||||||||
| Table 1: Several metal/metalloid contaminants of concern in groundwater. (Revised from[6]). | |||||||||||
Behavior in Groundwater
Dissolved metal behavior in groundwater is controlled by reactions within the aqueous phase and reactions between the contaminant metal and the solid phase (such as clay particles or sand grains in the aquifer). Hence, the chemical composition of the water and the aquifer mineralogy are important to metal contaminant behavior. In turn, the water chemistry can be controlled by the aquifer mineralogy, the chemical composition of the infiltrating plume, or reactions between the infiltrating plume and aquifer minerals. If carbon and other nutrients are available, reactions controlling metal behavior can be influenced by microbial activity. All metal contaminants in groundwater are distributed among dissolved species in which the metal ion (an ion is a dissolved species with a positive or negative charge) is associated with one or more ions, producing a different dissolved species with different chemical behavior[10][11]. For example, the mercury ion, Hg+2, readily associates with the chloride ion, Cl-, to produce a series of species (HgCl+, HgCl2°, HgCl3-) with concentrations dependent on the chemical composition of the groundwater. The collection of species is called the aqueous speciation of the contaminant metal. Some contaminants have a simple aqueous speciation and few species dominate over a broad range of groundwater conditions. Others have a complicated aqueous speciation that changes with pH, redox potential, and/or the presence or absence of certain ions. Uranium is an example of a complicated aqueous speciation in common aquifer conditions (Fig. 1). The aqueous speciation strongly influences sorption and precipitation reactions involving the contaminant metal, and thus influences the mobility of the metal in the aquifer and the methods of effective remediation (see article on Mobility of Metals and Metalloids).
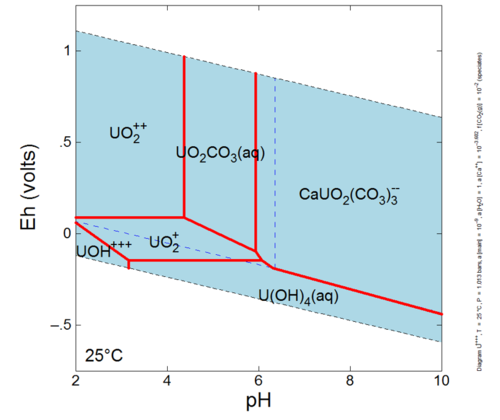
The most important aspect of a metal contaminants aqueous speciation is whether multiple oxidation states can occur over the range of conditions found in groundwater. Contaminants that have multiple oxidation states that are stable in groundwater are considered “redox sensitive”. Different oxidation states of a metal can have very different behaviors and toxicity in groundwater. For example, chromium in a plus six oxidation state (chromate) is more mobile and toxic than chromium in a plus three oxidation state. Table 2 lists whether several common metal contaminants are redox sensitive and the relative complexity of their aqueous speciation. Here, a complicated aqueous speciation is defined as having 4 or more species that dominate various portions of the field defined by a pH range of 4 to 10 and an Eh (Eh is a measure of the tendency of electrons to flow from one ion to another[13] that covers the stability field of water (as determined using the thermodynamic database “thermo_minteq” and The Geochemist’s Workbench® with [Cl-] and [SO4-2] = 10 mg/L). The complexity of aqueous speciation is not determined by redox sensitivity because many metals that do not change oxidation states with varying redox conditions may still form aqueous sulfide or bisulfide species under sufficiently reducing conditions.
The importance of aqueous speciation of a contaminant metal to environmental remediation can be illustrated by the behavior of uranium. Many groundwaters contaminated by uranium contain dissolved oxygen and moderate concentrations of dissolved carbon dioxide. Under these conditions, the dominant aqueous species of oxidized uranium [U(VI)] are neutrally or negatively charged carbonate species (Fig. 1). These species tend to keep oxidized uranium in the dissolved state, making it difficult to limit its mobility by causing it to adsorb or precipitate in a solid phase. The challenge is to find some method that either overcomes the effects of the carbonate species or changes the speciation of the uranium.
One method that has been studied extensively is to change the speciation by converting the uranium to the reduced form, U(IV), causing the uranium to precipitate as a low solubility U(IV) oxide. This can be done with chemicals (abiotic reduction) or by stimulating indigenous microbes to consume all of the oxygen in the groundwater (bioreduction). A common problem with this approach is that the groundwater naturally contains oxygen, and oxygenated groundwater will quickly flow into the treated zone re-oxidizing and remobilizing the uranium.
At some sites, oxidized uranium has been successfully treated with phosphate minerals because, under the conditions of the site groundwater, U(VI) is bound more strongly in the phosphate minerals than it is in the aqueous carbonate complexes. There are other ways to overcome the effects of the carbonate complexes and limit the mobility of uranium. The key idea to selecting a uranium remediation method that is likely to be successful, is understanding how the aqueous speciation of uranium will affect the treatment.
References
- ^ Fabian, G. and Watts, K., 2005. Army small arms training range environmental best practices (BMPs) Manual. DTC Project No. 9-CO-160-000-504, U.S. Army Environmental Center, Aberdeen Proving Ground, MD. Report pdf
- ^ United States Government Accountability Office (GAO), 2005. Groundwater contamination – DOD uses and develops a range of remediation technologies to clean up military sites. Report to congressional committees, GAO-05-666. Report pdf
- ^ Hering, J.G., Burris, D., Reisinger, H.J., O’Day, P., 2008. Environmental fate and exposure assessment for arsenic in groundwater. SERDP Project ER-1374. ER-1374
- ^ Riley, R.G. and Zachara, J.M., 1992. Chemical contaminants on DOE lands and selection of contaminant mixtures for subsurface science research. U.S. Department of Energy (No. DOE/ER--0547T). Report pdf
- ^ Hazen, T.C., Faybishenko, B., Jordan, P., 2008. Complexity of Groundwater Contaminants at DOE Sites. LBNL-4117E, Lawrence Berkeley National Laboratory. Report pdf
- ^ 6.0 6.1 United States Geological Survey (USGS). Contaminants found in groundwater. USGS Website
- ^ World Health Organization (WHO), 2016. Ten chemicals of major public health concern. [http://www.who.int/ipcs/assessment/public_health/chemicals_phc/en/ WHO Website[
- ^ U.S. Environmental Protection Agency (USEPA), 2016. Table of Regulated Drinking Water Contaminants.Table of Regulated Drinking Water
- ^ U.S. Environmental Protection Agency (USEPA), 2016. Secondary drinking water standards: guidance for nuisance chemicals. Webpage
- ^ Langmuir, D., 1997, Aqueous Environmental Geochemistry. Prentice-Hall, Inc. Upper Saddle River, NJ ISBN: 978-0023674129.
- ^ Drever, J.I., The Geochemistry of Natural Waters: Surface and Groundwater Environments. Prentice-Hall, Inc., ISBN 0132727900.
- ^ Bethke, C.M. and S. Yeakel, 2015. The geochemist’s workbench®, Release 10.0. Latest version available at [www.gwb.com www.gwb.com]
- ^ Hem, J.D., 1970. Chemical behavior of mercury in aqueous media. In Mercury in the Environment (Vol. 713, pp. 19-24). Washington, DC: US Government Printing Office. Report pdf