Difference between pages "1,4-Dioxane" and "NAPL Mobility"
m |
m |
||
| Line 1: | Line 1: | ||
| − | [[ | + | [[Wikipedia: Light non-aqueous phase liquid | Light non-aqueous phase liquid (LNAPL)]] source zones evolve over time, from a short period of expanding LNAPL bodies, to mature sites where LNAPL mobility becomes much lower or zero. The recent development of [[Wikipedia:LNAPL transmissivity | LNAPL transmissivity]] testing provides an improved metric for understanding whether mobile LNAPL can be effectively recovered from hydrocarbon sites. |
<div style="float:right;margin:0 0 2em 2em;">__TOC__</div> | <div style="float:right;margin:0 0 2em 2em;">__TOC__</div> | ||
| − | '''Related Article(s) | + | '''Related Article(s)''': |
| + | *LNAPL Source Zone Conceptual Models (Coming soon) | ||
| + | *LNAPL Remediation Technologies (Coming soon) | ||
| − | |||
| − | |||
| − | '''CONTRIBUTOR(S):''' [[ | + | '''CONTRIBUTOR(S):''' [[Andrew Kirkman]] |
| − | '''Key Resource(s)''': | + | '''Key Resource(s)''': |
| + | *[https://lnapl-3.itrcweb.org LNAPL Site Management: LCSM Evolution, Decision Process, and Remedial Technologies]<ref name= "ITRC2018">ITRC, 2018. LNAPL Site Management: LCSM evolution, decision process, and remedial technologies (LNAPL-3). Interstate Technical and Regulatory Council https://lnapl-3.itrcweb.org/</ref>. Section 3 provides an introductory discussion of LNAPL mobility concepts. | ||
| + | *[https://www.api.org/oil-and-natural-gas/environment/clean-water/ground-water/lnapl/ldrm API LNAPL Distribution and Recovery Model (LDRM), API 4760] <ref name= "Charbeneau2007">Charbeneau, R.J., 2007. LNAPL Distribution and Recovery Model. Distribution and Recovery of Petroleum Hydrocarbon Liquids in Porous Media. Vol. 1. API Publication 4760. [[media:API2007_LDRM.pdf | Report.pdf]] https://www.api.org/oil-and-natural-gas/environment/clean-water/ground-water/lnapl/ldrm</ref>. Provides detailed discussion of LNAPL mobility as related to saturation, soil properties and capillary pressure. | ||
| + | *[https://www.crccare.com/files/dmfile/CRCCARETechnicalreport44_TechnicalmeasurementguidanceforLNAPLnaturalsourcezonedepletion.pdf Technical measurement guidance for LNAPL natural source zone depletion]<ref name= "CRCCARE2018">CRC CARE, 2018. Technical measurement guidance for LNAPL natural source zone depletion. Cooperative Research Centre for Contamination Assessment and Remediation of the Environment, Newcastle, Australia. Technical Report no. 44. 254p [[media:CRCCARE2018_Measurement_Guidance_for_LNAPL_NSZD.pdf | Report.pdf]] https://www.crccare.com/files/dmfile/CRCCARETechnicalreport44_TechnicalmeasurementguidanceforLNAPLnaturalsourcezonedepletion.pdf</ref>. Section 6 provides a discussion of compositional changes to LNAPL over time and how to use those changes to calculate natural source zone depletion rates. | ||
| − | + | ==Introduction== | |
| + | LNAPL can be found in the subsurface in three different states: | ||
| + | #'''Residual LNAPL''' is trapped and immobile but can undergo composition change and generate dissolved hydrocarbon plumes and/or vapor sources in the unsaturated zones. | ||
| + | #'''Mobile LNAPL''' is LNAPL at greater than the residual saturation which can therefore accumulate in a well where it is potentially recoverable. However, in this state LNAPL saturation is insufficient to drive migration (i.e., the LNAPL body is not expanding). | ||
| + | #'''Migrating LNAPL''' is LNAPL at greater than the residual saturation that expands into previously unimpacted locations over time (e.g., LNAPL appears in a monitoring well that had no previous detections). | ||
| − | == | + | When released to the subsurface, light non-aqueous phase liquids form source zones which evolve over time with changing impacts to soil, groundwater, and at some sites surface water. Shallow unsaturated soils may also be impacted by intrusion of vapors released by the LNAPL. (See [[Vapor Intrusion - Separation Distances from Petroleum Sources]].) LNAPL mobility is typically highest during the time of the release to the environment. Once the release stops, LNAPL mobility and the potential for migration rapidly decrease. The LNAPL body may initially contain mobile LNAPL near the release point. However, as the LNAPL body spreads, the driving head and pressure gradient decrease, and the LNAPL thickness (a key factor in mobility) decreases. The LNAPL body also begins to weather, losing volatiles and biodegradable compounds. Over time, this weathering process alters the LNAPL composition and stabilizes the LNAPL body, and the fraction of LNAPL that remains mobile therefore decreases<ref name= "ITRC2018"/><ref name= "CRCCARE2018"/><ref>Mahler, N., Sale, T. and Lyverse, M., 2012. A mass balance approach to resolving LNAPL stability. Groundwater, 50(6), pp.861-871. [https://doi.org/10.1111/j.1745-6584.2012.00949.x [10.1111/j.1745-6584.2012.00949.x 10.1111/j.1745-6584.2012.00949.x]</ref>. LNAPL bodies from historical releases are dominated by immobile residual LNAPL. |
| − | + | ||
| − | + | Because LNAPL bodies evolve over time, LNAPL mobility is a much more significant concern during and immediately after the release period than it is after the release is halted. Tools are now available to assess the magnitude of LNAPL mobility at individual release sites to support decision making regarding potential remedial actions. One such tool is the American Petroleum Institute’s [https://www.api.org/oil-and-natural-gas/environment/clean-water/ground-water/lnapl/ldrm LNAPL Distribution and Recovery Model (LDRM)] which is available as a free download. Please see the LNAPL Mobility and Recovery section below for a more complete discussion of modeling tools. | |
| − | {| class="wikitable" style="float:right; margin-left: 10px;" | + | |
| − | |+Table 1. | + | In order to understand LNAPL mobility, key concepts from the science of multiphase flow in porous media (e.g. soil and aquifer material) are important. Consider dry soil to which a small amount of water is added. The open pore space in the soil, initially filled with 100 percent air, now contains a mix of air and water. Therefore, a soil can have more than one fluid within the pores (“multiphase”). The fraction of pore space filled with a given fluid is termed “saturation”. The air saturation of the initially dry soil was 100% and the water saturation was zero. When considering a petroleum release site, three different fluids may exist in the pore space: air, water and LNAPL. The sum of the saturation of the three fluids is 100%. All three fluids can exist within the subsurface with varying saturations. |
| + | |||
| + | ==LNAPL Mobility Conceptualized== | ||
| + | [[File:Kirkman1w2Fig1.png|thumb|left|Figure 1. LNAPL Distribution in a research sand tank<ref>Hawkins, A. M., 2013. Processes Controlling the behavior of LNAPLs at groundwater surface water interfaces, Master of Science Thesis, Colorado State University, Ft. Collins, Colorado. [[media:Hawkins2013_LNAPL_at_GW_SW_Interfaces.pdf | Report.pdf]]</ref>.]] | ||
| + | Figure 1 shows a research tank with a well screen in a porous media (sand) containing LNAPL. The LNAPL fluoresces under ultraviolet (UV) light, while water and air do not. The column of yellow fluorescence in the well represents the gauged LNAPL thickness and corresponds to the vertical interval over which LNAPL flows in the formation<ref name= "Huntley2000">Huntley, D., 2000. Analytic determination of hydrocarbon transmissivity from baildown tests. Groundwater, 38(1), pp.46-52. [https://doi.org/10.1111/j.1745-6584.2000.tb00201.x doi: 10.1111/j.1745-6584.2000.tb00201.x]</ref>. The yellow fluorescence to the left of the well represents LNAPL occurring within soil pores. Below the potentiometric surface line, the pores are dominated by water (dark pores) and LNAPL. Above this line LNAPL and water still exist in pores, but the air saturation increases with elevation. The incomplete yellow fluorescence in the formation illustrates how water and LNAPL occupy pores over the mobile interval. This occurs because the mobile LNAPL cannot displace all of the water from the pores. | ||
| + | |||
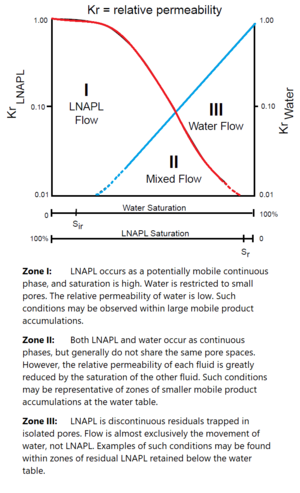
| + | [[File:Kirkman1w2Fig2.png|thumb|Figure 2. Hypothetical relative permeability curves for water and an LNAPL in a porous medium<ref name= "USEPA1996"/><ref>Newell, C. J., Acree*, S. D., Ross, R.R., and Huling, S.G. 1995. Light non-aqueous phase liquid. U.S. Environmental Protection Agency, Washington, DC. EPA/540/S-95/500 (NTIS 95-267738) [[media:Newell1995_EPA_540_S-95_500_lnapl.pdf | Report.pdf]]</ref><ref>Williams, D.E. and Wilder, D.G., 1971. Gasoline Pollution of a Ground‐Water Reservoir—A Case History. Groundwater, 9(6), pp.50-56. [https://doi.org/10.1111/j.1745-6584.1971.tb03577.x doi: 10.1111/j.1745-6584.1971.tb03577.x]</ref>.]] | ||
| + | LNAPL mobility can be described mathematically by Darcy’s Law, an empirically derived equation describing the flow of fluids through porous media where the specific discharge, ''q'', is equal to the product of the hydraulic conductivity, ''K'', and the hydraulic gradient, ''i''. While this is the same Darcy’s Law that is used to describe groundwater flow (see [[Advection and Groundwater Flow]]), there is an important additional term, Relative Permeability, that is included when using Darcy’s Law to describe the flow of LNAPL in the subsurface. Relative permeability is the ratio of the effective permeability of a fluid at a specified saturation to the intrinsic permeability of the medium at 100-percent saturation<ref>Mercer, J.W. and Cohen, R.M., 1990. A review of immiscible fluids in the subsurface: properties, models, characterization and remediation. Journal of contaminant hydrology, 6(2), pp.107-163. [https://doi.org/10.1016/0169-7722(90)90043-G doi: 10.1016/0169-7722(90)90043-G]</ref>. The USEPA (1996)<ref name= "USEPA1996">USEPA, 1996. How to effectively recover free product at leaking underground storage sites. A guide for state regulators. USEPA 510-R-96-001. U.S. Environmental Protection Agency, 162 pp. [[media:USEPA1996_510_R-96_001_Recover_Free_Prod_at_LUSTS.pdf| Report.pdf]]</ref> describes relative permeability this way: | ||
| + | |||
| + | <q>''The relative permeability of a particular geologic media that is completely saturated with a particular fluid is equal to the intrinsic permeability. When more than one fluid (i.e., air, water, petroleum hydrocarbon) exists in a porous medium, the fluids compete for pore space thereby reducing the relative permeability of the media and the mobility of the fluid. This reduction can be quantified by multiplying the intrinsic permeability of the geologic media by the relative permeability. As with saturation, the mobility of each fluid phase present varies from zero (0% saturation) to one (100% saturation).''</q> | ||
| + | |||
| + | Figure 2 shows an example of relative permeability curves for a water-LNAPL system. The curves representing water saturation (starting at top right, blue line) and hydrocarbon saturation (starting at top left, red line) are contrary to one another and divide the figure into three flow zones. Zone I, where hydrocarbon saturation is relatively high, is dominated by hydrocarbon flow. Water saturation is relatively high in Zone III, and therefore water flow is dominant. Flow of both water and LNAPL characterizes Zone II. | ||
| + | |||
| + | The relative permeability term scales the LNAPL conductivity to the fraction of pores occupied by LNAPL. If LNAPL occupies 50 percent of the pores, then it will only flow through LNAPL-filled pores and therefore does not use the entire permeability of the soil. If the sum of the pores is considered a pipe, then a pipe half full will transmit less LNAPL than a pipe completely full for a given pressure gradient. The relative permeability term is used to account for this effect. | ||
| + | |||
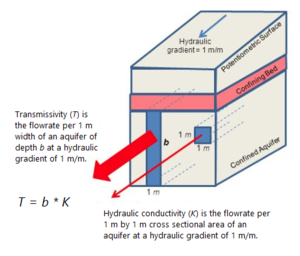
| + | [[File:Kirkman1w2Fig3.png|thumb|left|Figure 3. Aquifer transmissivity versus hydraulic conductivity.]] | ||
| + | LNAPL saturation varies with elevation across the gauged LNAPL thickness, where LNAPL conductivity is lowest near the oil/water interface and highest near the air/LNAPL interface. To provide a single metric for LNAPL mobility, the conductivity must be summed across the mobile LNAPL interval to give the LNAPL transmissivity. | ||
| + | |||
| + | LNAPL transmissivity has been identified as a key metric for evaluating the likely effectiveness of direct LNAPL recovery compared to traditional measurements such as gauged LNAPL thickness in monitoring wells<ref>Kolhatkar, R., Kremesec, V., Rubin, S., Yukawa, C. and Senn, R., 1999. Application of field and analytical techniques to evaluate recoverability of subsurface free phase hydrocarbons. Petroleum Hydrocarbons and Organic Chemicals in Ground Water, pp.5-15.</ref><ref>Lundy, D.A. and Zimmerman, L.M., 1996, May. Assessing the recoverability of LNAPL plumes for recovery system conceptual design. In Proceedings of the 10th National Outdoor Action Conference and Expo (pp. 13-15).</ref><ref name= "Huntley2000"/><ref name= "Kirman2013">Kirkman, A.J., 2013. Refinement of Bouwer‐Rice baildown test analysis. Groundwater Monitoring & Remediation, 33(1), pp.105-110. [https://doi.org/10.1111/j.1745-6592.2012.01411.x doi: 10.1111/j.1745-6592.2012.01411.x]</ref><ref name= "ASTM2013">ASTM, 2013. ASTM E2856-13, Standard guide for estimation of LNAPL transmissivity, ASTM International, West Conshohocken, PA.</ref><ref name= "Charbeneau2016">Charbeneau, R., Kirkman, A., and Muthu, R., 2016. API LNAPL Transmissivity Workbook: A Tool for Baildown Test Analysis – User Guide. American Petroleum Institute Publication 4762.</ref><ref name= "Palmier2016">Palmier, C., Dodt, M. and Atteia, O., 2016. Comparison of Oil Transmissivity Methods Using Bail‐Down Test Data. Groundwater Monitoring & Remediation, 36(3), pp.73-83.[[media:Palmier2016_LNAPLBaildownTesting.pdf| Report.pdf]]</ref><ref>Lenhard, R.J., Rayner, J.L. and Davis, G.B., 2017. A practical tool for estimating subsurface LNAPL distributions and transmissivity using current and historical fluid levels in groundwater wells: Effects of entrapped and residual LNAPL. Journal of contaminant hydrology, 205, pp.1-11. [[media:Lenhard2017_Estimating_Subsurface_LNAPL.pdf | Report.pdf]]</ref>. LNAPL transmissivity represents the volume of LNAPL flow a formation can produce per unit time over a unit width for a unit hydraulic gradient. Aquifer transmissivity is the primary metric for evaluating how much water can be produced from a water bearing unit in the subsurface and accounts for the thickness of the saturated aquifer and the ease with which the aquifer can transmit a given fluid per unit volume. The concept of transmissivity is illustrated in Figure 3. It is reasonable to apply similar thinking to estimation of the amount of LNAPL that could be produced by a particular subsurface matrix, and therefore transmissivity has become a key metric used to predict whether direct recovery (direct pumping) of LNAPL is likely to be effective. | ||
| + | |||
| + | The average LNAPL recovery rate was shown to be closely related to the average LNAPL transmissivity as well as to the LNAPL thickness in a series of 10-hour pump tests of three wells at an LNAPL-impacted site (Table 1, ITRC 2018)<ref name= "ITRC2018"/>. The measured LNAPL thickness in the source well (MW-1) was 3.7 times higher than in the LNAPL fringe well (MW-3), while the ratio of their average LNAPL transmissivities was 5. The average recovery rate was 12 times greater at the source well than at the fringe well. In this study, LNAPL thickness, average transmissivity and average recovery rate were all strongly correlated, with correlation coefficients of 0.94 and above. | ||
| + | {| class="wikitable" style="float:right; margin-left:10px;text-align:center;" | ||
| + | |+ Table 1. LNAPL Transmissivity Results from One LNAPL Site<ref name= "ITRC2018"/> | ||
|- | |- | ||
| − | ! | + | ! Well ID |
| − | ! | + | ! Well Location |
| − | ! | + | ! LNAPL Thickness in Well<br/>(ft) |
| + | ! Average LNAPL Transmissivity<br/>(ft<sup>2</sup>/day) | ||
| + | ! Average LNAPL Recovery Rate<br/>(gallons/minute) | ||
|- | |- | ||
| − | | | + | | '''MW-1''' || Source || 8.13 || 100 || 1.2 |
|- | |- | ||
| − | | | + | | '''MW-2''' || Mid-point || 4.39 || 60 || 0.3 |
|- | |- | ||
| − | | | + | | '''MW-3''' || LNAPL || 2.2 || 20 || 0.1 |
|- | |- | ||
| − | | | + | | colspan="2" | '''Ratio of MW-1 to MW-3''' || 3.7 || 5.0 || 12 |
| − | |||
| − | |||
| − | |- | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | | | ||
| − | |||
|} | |} | ||
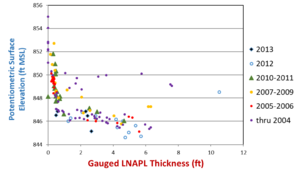
| − | + | An example in which LNAPL transmissivity would be better than LNAPL thickness at predicting LNAPL recoverability is shown in Figure 4. At this site there was little or no change in LNAPL thickness over 10 years of LNAPL recovery using a vacuum truck. If the LNAPL thickness were affected by the ongoing recovery action, a clear trend of decreasing LNAPL thickness over time would have been observed, which would have resulted in clustering of early time data on the right side of the graph and late time data on the left<ref name= "ITRC2018"/>. | |
| + | [[File:Kirkman1w2Fig4.png|thumb|left|Figure 4. Gauged LNAPL thickness versus potentiometric surface elevation through time<ref name= "ITRC2018"/><ref name= "Kirkman2_2013">Kirkman, A.J., Adamski, M. and Hawthorne, J.M., 2013. Identification and assessment of confined and perched LNAPL conditions. Groundwater Monitoring & Remediation, 33(1), pp.75-86. [https://doi.org/10.1111/j.1745-6592.2012.01412.x doi: 10.1111/j.1745-6592.2012.01412.x]</ref>), showing 10 years of LNAPL recovery with little to no change in LNAPL thickness.]] | ||
| − | + | Based on the practical experience from five LNAPL sites, the ITRC proposed a threshold LNAPL transmissivity for recoverability: values below 0.1 to 0.8 ft<sup>2</sup>/day indicate low recoverability. Therefore, the majority of the LNAPL at a site in this category is in a state of lesser mobility, and the remaining source is dominated by residual saturation <ref name= "ITRC2018"/>. For the five LNAPL sites, the ITRC concluded: “These sites were closed or granted no further action after developing comprehensive LNAPL Conceptual Site Models and operating LNAPL recovery systems. All sites demonstrated achievement of the impracticable limit (i.e., the lack of LNAPL recoverability) irrespective of in-well LNAPL thickness remaining. Since that time, data from four additional sites, with comprehensive LCSMs based on high-resolution data, were analyzed and affirmed the proposed limits.” The ITRC (2018)<ref name= "ITRC2018"/> recently expanded the technical basis for this important LNAPL transmissivity threshold with more technical background and additional case studies. | |
| − | + | ==Characterization of LNAPL Mobility== | |
| + | Field testing for LNAPL transmissivity transforms the gauged LNAPL thickness, fluid parameters of density and viscosity and the relative permeability into a single value. As saturation decreases, the interconnectivity of the pores decreases, and the ability of the soil to transmit LNAPL for a given gradient decreases. | ||
| − | + | LNAPL transmissivity can be estimated with LNAPL baildown or other well testing techniques where the well screen intersects the entire interval of mobile LNAPL. ASTM (2013)<ref name= "ASTM2013"/> and Gatsios et al. (2018)<ref>Gatsios, E., García-Rincón, J., Rayner, J.L., McLaughlan, R.G. and Davis, G.B., 2018. LNAPL transmissivity as a remediation metric in complex sites under water table fluctuations. Journal of Environmental Management, 215, pp.40-48. [[media:Gatsios2018_LNAPL_Transmissivity_as_Metric.pdf | Report.pdf]]</ref> describe four methods: | |
| − | |||
| − | + | *Short-Term Baildown Tests: LNAPL is quickly removed from a well, and data showing how quickly LNAPL refills the well are collected. These data are analyzed using methods similar to analyzing slug tests. An easy to use spreadsheet<ref name= "Charbeneau2016"/> is available [https://www.api.org/oil-and-natural-gas/environment/clean-water/ground-water/lnapl/transmissivity-workbook here] to help process the data generated by short-term baildown tests and obtain an LNAPL transmissivity estimate. Short-term baildown tests are the most commonly used method. | |
| + | *Manual Skimming Method: LNAPL is repeatedly recovered over time (on the order of several days), gauging the well as it recovers, and then initiating another recovery event before the LNAPL level rebounds by more than 25% of the original drawdown. This is done until the LNAPL removal rate stabilizes, and then this rate is used to calculate transmissivity. | ||
| + | *Long-Term Recovery-Based Methods: Typically applied to sites with on-going LNAPL recovery. There are several equations which can be used to calculate LNAPL transmissivity from the long-term recovery data. | ||
| + | *Tracer-Based Method: An emerging, less common method involving placing fluorescent dyes that are soluble in LNAPL but not water into a well. The dye concentration is measured over time to calculate the LNAPL flux through the well. | ||
| − | + | Two comparative studies have shown that different LNAPL transmissivity measurement techniques yield similar results<ref name= "Palmier2016"/><ref name= "Kirman2013"/>). For sites with more complex conditions such as perched LNAPL, confined LNAPL, and changes in potentiometric surface, see ASTM Standard E2856-13<ref name= "ASTM2013"/> and Kirkman et al. (2013)<ref name= "Kirkman2_2013"/>. | |
| − | |||
| − | + | In addition to field testing, LNAPL transmissivity can also be estimated using an appropriate computer-based model, although it is best to calibrate the model to observed field values initially. Please see LNAPL Mobility and Recovery Models (below) for more on this topic. | |
| − | + | ==Why is LNAPL mobility important?== | |
| + | All LNAPL starts out mobile during the release and the resulting pore-invading process, often referred to as “drainage” conditions (because as the LNAPL enters a pore, the water or air is “drained” out). LNAPL must displace fluids such as water and air to invade the pores. While larger pores in a given soil can be easily entered by LNAPL that has relatively low driving [[Wikipedia: Hydraulic head | head]] (pressure) relative to water or air, smaller pores require larger heads relative to those fluids. This behavior results in incomplete LNAPL saturation of the pores, as shown in Figure 1. | ||
| − | + | Concerns with LNAPL mobility are primarily focused on preventing additional migration and on reduction of the source volume. Consider a release of LNAPL to the subsurface. During the invasion process LNAPL is migrating down to the water table while spreading. Pores at the front of the migration are interconnected as saturation is increasing. Hence all LNAPL is mobile. Once the release stops, pores near the release point and in the center of the LNAPL body will have more LNAPL flowing out of them than is flowing in. LNAPL saturations decrease as pores are evacuated of LNAPL. During this evacuation (also referred to as “imbibition”) the pores become disconnected and some LNAPL is left in smaller, now disconnected, pores. This phenomenon is due to capillary forces inhibiting gravity drain/hydraulic gradient mechanisms. This is similar to the fraction of water that will not drain out of a sponge. This behavior, a key feature of multiphase flow in porous media, results in incomplete removal of LNAPL from the soil and the formation of residual LNAPL (which can be conceptualized as a disconnected single LNAPL blob in a single pore). The fraction of LNAPL retained is termed “residual LNAPL saturation” and is a function of the pore size distribution or the soil type, the maximum saturation LNAPL initially achieved and the water saturation. | |
| − | + | Multiple researchers<ref>Lenhard, R.J., Oostrom, M. and Dane, J.H., 2004. A constitutive model for air-NAPL-water flow in the vadose zone accounting for immobile, non-occluded (residual) NAPL in strongly water-wet porous media. Journal of Contaminant Hydrology, Vol. 73(1-4), pp 283-304 [[media:Lenhard2004_air-NAPL-water_flow.pdf| Report.pdf]]</ref><ref name= "Johnston2005">Johnston, C.D. and Adamski, M., 2005, August. Relationship between initial and residual LNAPL saturation for different soil types. In Proceedings of the 2005 Petroleum Hydrocarbons and Organic Chemicals in Groundwater®: Prevention, Assessment, and Remediation Conference (pp. 17-19). [[media:Johnston2005_Residual_LNAPL_Saturation.pdf| Report.pdf]]</ref> have investigated the range of expected values of residual saturation in soils at LNAPL release sites. The overall conclusion of this work is that residual saturation depends on the amount of LNAPL initially present in the pores (initial saturation, S<sub>ni</sub>), the soil type and the water saturation. Figure 5 illustrates how residual saturation values in the vadose zone vary with soil type and initial LNAPL saturation<ref name= "Johnston2005"/>. Key points from Figure 5 include: a) finer-grained soils like silts (ML) and clays (CL) often have higher residual saturation values than sands (SC or SP); and b) higher initial saturation values result in higher residual saturation values. | |
| − | + | ==LNAPL Mobility and Recovery Models== | |
| + | LNAPL distribution and mobility can be evaluated using public domain computer models. The LNAPL Distribution and Recovery Model estimates LNAPL transmissivity, saturation and overall volume in the subsurface and aids in assessing the potential effectiveness of recovery efforts to reduce LNAPL source volume. The Hydrocarbon Spill Screening Model is a US EPA model for evaluating the infiltration of LNAPL following a release and the resulting risks as LNAPL reaches the water-table and spreads laterally. | ||
| − | + | *[https://www.api.org/oil-and-natural-gas/environment/clean-water/ground-water/lnapl/ldrm API LNAPL Distribution and Recovery Model (LDRM), API 4760]<ref name= "Charbeneau2007"/> – Addresses the recoverability of LNAPL in terms of time, induced drawdown, well spacing and residual fraction. | |
| − | + | *[https://www.epa.gov/water-research/hydrocarbon-spill-screening-model-hssm-windows-version Hydrocarbon Spill Screening Model (HSSM)]<ref>Weaver, J.W., Charbeneau, R.J., Tauxe, J.D., Lien, B.K. and Provost, J.B., 1995. The Hydrocarbon spill screening model (HSSM) Volume 1: User’s Guide. US EPA, publication EPA/600/R-94/039a, 229pp. [[media:Weaver1995_HSSMv1.pdf | Report.pdf]]</ref> – Evaluates potential risks of releases and promotes continuing improvement towards prevention. | |
| − | + | ==LNAPL Mobility and Risk == | |
| + | Residual LNAPL poses the same risk of groundwater contamination and [[Vapor Intrusion - Separation Distances from Petroleum Sources | petroleum vapor intrusion]] as mobile LNAPL<ref name= "ITRC2018"/>. The LNAPL composition, not the LNAPL thickness or saturation in soil, drives the concentrations in vapor and groundwater phases and therefore the risk to receptors. Once most of the LNAPL is immobile and residual LNAPL dominates the source area, removal of any small remaining mobile fraction is not expected to reduce risks of groundwater contamination or petroleum vapor intrusion in a meaningful way. | ||
| − | + | ==Summary and Key Conclusions== | |
| + | Mobile LNAPL can pose a risk where continued migration and expansion of the LNAPL footprint is occurring. LNAPL recovery efforts should focus on sites where LNAPL migration has the potential to expand the LNAPL footprint and where LNAPL recovery can significantly reduce the overall source volume. Where most of the LNAPL is immobile, recovery of a small amount of mobile LNAPL is not expected to substantially reduce risks of groundwater contamination or petroleum vapor intrusion. | ||
| − | + | The ITRC (2018)<ref name= "ITRC2018"/> discusses lines of evidence for evaluating LNAPL Migration risk as well as the benefits of LNAPL recovery in source reduction. Often LNAPL sites exhibiting low transmissivity values (i.e. 0.1 to 0.8 ft<sup>2</sup>/day) or with stable LNAPL bodies that are comprised of mostly residual LNAPL will not benefit from recovery attempts to remove mobile LNAPL. | |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
==References== | ==References== | ||
| − | + | <references/> | |
| − | <references /> | ||
==See Also== | ==See Also== | ||
| − | |||
| − | |||
| − | |||
Revision as of 11:31, 6 February 2020
Light non-aqueous phase liquid (LNAPL) source zones evolve over time, from a short period of expanding LNAPL bodies, to mature sites where LNAPL mobility becomes much lower or zero. The recent development of LNAPL transmissivity testing provides an improved metric for understanding whether mobile LNAPL can be effectively recovered from hydrocarbon sites.
Related Article(s):
- LNAPL Source Zone Conceptual Models (Coming soon)
- LNAPL Remediation Technologies (Coming soon)
CONTRIBUTOR(S): Andrew Kirkman
Key Resource(s):
- LNAPL Site Management: LCSM Evolution, Decision Process, and Remedial Technologies[1]. Section 3 provides an introductory discussion of LNAPL mobility concepts.
- API LNAPL Distribution and Recovery Model (LDRM), API 4760 [2]. Provides detailed discussion of LNAPL mobility as related to saturation, soil properties and capillary pressure.
- Technical measurement guidance for LNAPL natural source zone depletion[3]. Section 6 provides a discussion of compositional changes to LNAPL over time and how to use those changes to calculate natural source zone depletion rates.
Introduction
LNAPL can be found in the subsurface in three different states:
- Residual LNAPL is trapped and immobile but can undergo composition change and generate dissolved hydrocarbon plumes and/or vapor sources in the unsaturated zones.
- Mobile LNAPL is LNAPL at greater than the residual saturation which can therefore accumulate in a well where it is potentially recoverable. However, in this state LNAPL saturation is insufficient to drive migration (i.e., the LNAPL body is not expanding).
- Migrating LNAPL is LNAPL at greater than the residual saturation that expands into previously unimpacted locations over time (e.g., LNAPL appears in a monitoring well that had no previous detections).
When released to the subsurface, light non-aqueous phase liquids form source zones which evolve over time with changing impacts to soil, groundwater, and at some sites surface water. Shallow unsaturated soils may also be impacted by intrusion of vapors released by the LNAPL. (See Vapor Intrusion - Separation Distances from Petroleum Sources.) LNAPL mobility is typically highest during the time of the release to the environment. Once the release stops, LNAPL mobility and the potential for migration rapidly decrease. The LNAPL body may initially contain mobile LNAPL near the release point. However, as the LNAPL body spreads, the driving head and pressure gradient decrease, and the LNAPL thickness (a key factor in mobility) decreases. The LNAPL body also begins to weather, losing volatiles and biodegradable compounds. Over time, this weathering process alters the LNAPL composition and stabilizes the LNAPL body, and the fraction of LNAPL that remains mobile therefore decreases[1][3][4]. LNAPL bodies from historical releases are dominated by immobile residual LNAPL.
Because LNAPL bodies evolve over time, LNAPL mobility is a much more significant concern during and immediately after the release period than it is after the release is halted. Tools are now available to assess the magnitude of LNAPL mobility at individual release sites to support decision making regarding potential remedial actions. One such tool is the American Petroleum Institute’s LNAPL Distribution and Recovery Model (LDRM) which is available as a free download. Please see the LNAPL Mobility and Recovery section below for a more complete discussion of modeling tools.
In order to understand LNAPL mobility, key concepts from the science of multiphase flow in porous media (e.g. soil and aquifer material) are important. Consider dry soil to which a small amount of water is added. The open pore space in the soil, initially filled with 100 percent air, now contains a mix of air and water. Therefore, a soil can have more than one fluid within the pores (“multiphase”). The fraction of pore space filled with a given fluid is termed “saturation”. The air saturation of the initially dry soil was 100% and the water saturation was zero. When considering a petroleum release site, three different fluids may exist in the pore space: air, water and LNAPL. The sum of the saturation of the three fluids is 100%. All three fluids can exist within the subsurface with varying saturations.
LNAPL Mobility Conceptualized

Figure 1 shows a research tank with a well screen in a porous media (sand) containing LNAPL. The LNAPL fluoresces under ultraviolet (UV) light, while water and air do not. The column of yellow fluorescence in the well represents the gauged LNAPL thickness and corresponds to the vertical interval over which LNAPL flows in the formation[6]. The yellow fluorescence to the left of the well represents LNAPL occurring within soil pores. Below the potentiometric surface line, the pores are dominated by water (dark pores) and LNAPL. Above this line LNAPL and water still exist in pores, but the air saturation increases with elevation. The incomplete yellow fluorescence in the formation illustrates how water and LNAPL occupy pores over the mobile interval. This occurs because the mobile LNAPL cannot displace all of the water from the pores.
LNAPL mobility can be described mathematically by Darcy’s Law, an empirically derived equation describing the flow of fluids through porous media where the specific discharge, q, is equal to the product of the hydraulic conductivity, K, and the hydraulic gradient, i. While this is the same Darcy’s Law that is used to describe groundwater flow (see Advection and Groundwater Flow), there is an important additional term, Relative Permeability, that is included when using Darcy’s Law to describe the flow of LNAPL in the subsurface. Relative permeability is the ratio of the effective permeability of a fluid at a specified saturation to the intrinsic permeability of the medium at 100-percent saturation[10]. The USEPA (1996)[7] describes relative permeability this way:
The relative permeability of a particular geologic media that is completely saturated with a particular fluid is equal to the intrinsic permeability. When more than one fluid (i.e., air, water, petroleum hydrocarbon) exists in a porous medium, the fluids compete for pore space thereby reducing the relative permeability of the media and the mobility of the fluid. This reduction can be quantified by multiplying the intrinsic permeability of the geologic media by the relative permeability. As with saturation, the mobility of each fluid phase present varies from zero (0% saturation) to one (100% saturation).
Figure 2 shows an example of relative permeability curves for a water-LNAPL system. The curves representing water saturation (starting at top right, blue line) and hydrocarbon saturation (starting at top left, red line) are contrary to one another and divide the figure into three flow zones. Zone I, where hydrocarbon saturation is relatively high, is dominated by hydrocarbon flow. Water saturation is relatively high in Zone III, and therefore water flow is dominant. Flow of both water and LNAPL characterizes Zone II.
The relative permeability term scales the LNAPL conductivity to the fraction of pores occupied by LNAPL. If LNAPL occupies 50 percent of the pores, then it will only flow through LNAPL-filled pores and therefore does not use the entire permeability of the soil. If the sum of the pores is considered a pipe, then a pipe half full will transmit less LNAPL than a pipe completely full for a given pressure gradient. The relative permeability term is used to account for this effect.
LNAPL saturation varies with elevation across the gauged LNAPL thickness, where LNAPL conductivity is lowest near the oil/water interface and highest near the air/LNAPL interface. To provide a single metric for LNAPL mobility, the conductivity must be summed across the mobile LNAPL interval to give the LNAPL transmissivity.
LNAPL transmissivity has been identified as a key metric for evaluating the likely effectiveness of direct LNAPL recovery compared to traditional measurements such as gauged LNAPL thickness in monitoring wells[11][12][6][13][14][15][16][17]. LNAPL transmissivity represents the volume of LNAPL flow a formation can produce per unit time over a unit width for a unit hydraulic gradient. Aquifer transmissivity is the primary metric for evaluating how much water can be produced from a water bearing unit in the subsurface and accounts for the thickness of the saturated aquifer and the ease with which the aquifer can transmit a given fluid per unit volume. The concept of transmissivity is illustrated in Figure 3. It is reasonable to apply similar thinking to estimation of the amount of LNAPL that could be produced by a particular subsurface matrix, and therefore transmissivity has become a key metric used to predict whether direct recovery (direct pumping) of LNAPL is likely to be effective.
The average LNAPL recovery rate was shown to be closely related to the average LNAPL transmissivity as well as to the LNAPL thickness in a series of 10-hour pump tests of three wells at an LNAPL-impacted site (Table 1, ITRC 2018)[1]. The measured LNAPL thickness in the source well (MW-1) was 3.7 times higher than in the LNAPL fringe well (MW-3), while the ratio of their average LNAPL transmissivities was 5. The average recovery rate was 12 times greater at the source well than at the fringe well. In this study, LNAPL thickness, average transmissivity and average recovery rate were all strongly correlated, with correlation coefficients of 0.94 and above.
| Well ID | Well Location | LNAPL Thickness in Well (ft) |
Average LNAPL Transmissivity (ft2/day) |
Average LNAPL Recovery Rate (gallons/minute) |
|---|---|---|---|---|
| MW-1 | Source | 8.13 | 100 | 1.2 |
| MW-2 | Mid-point | 4.39 | 60 | 0.3 |
| MW-3 | LNAPL | 2.2 | 20 | 0.1 |
| Ratio of MW-1 to MW-3 | 3.7 | 5.0 | 12 | |
An example in which LNAPL transmissivity would be better than LNAPL thickness at predicting LNAPL recoverability is shown in Figure 4. At this site there was little or no change in LNAPL thickness over 10 years of LNAPL recovery using a vacuum truck. If the LNAPL thickness were affected by the ongoing recovery action, a clear trend of decreasing LNAPL thickness over time would have been observed, which would have resulted in clustering of early time data on the right side of the graph and late time data on the left[1].
Based on the practical experience from five LNAPL sites, the ITRC proposed a threshold LNAPL transmissivity for recoverability: values below 0.1 to 0.8 ft2/day indicate low recoverability. Therefore, the majority of the LNAPL at a site in this category is in a state of lesser mobility, and the remaining source is dominated by residual saturation [1]. For the five LNAPL sites, the ITRC concluded: “These sites were closed or granted no further action after developing comprehensive LNAPL Conceptual Site Models and operating LNAPL recovery systems. All sites demonstrated achievement of the impracticable limit (i.e., the lack of LNAPL recoverability) irrespective of in-well LNAPL thickness remaining. Since that time, data from four additional sites, with comprehensive LCSMs based on high-resolution data, were analyzed and affirmed the proposed limits.” The ITRC (2018)[1] recently expanded the technical basis for this important LNAPL transmissivity threshold with more technical background and additional case studies.
Characterization of LNAPL Mobility
Field testing for LNAPL transmissivity transforms the gauged LNAPL thickness, fluid parameters of density and viscosity and the relative permeability into a single value. As saturation decreases, the interconnectivity of the pores decreases, and the ability of the soil to transmit LNAPL for a given gradient decreases.
LNAPL transmissivity can be estimated with LNAPL baildown or other well testing techniques where the well screen intersects the entire interval of mobile LNAPL. ASTM (2013)[14] and Gatsios et al. (2018)[19] describe four methods:
- Short-Term Baildown Tests: LNAPL is quickly removed from a well, and data showing how quickly LNAPL refills the well are collected. These data are analyzed using methods similar to analyzing slug tests. An easy to use spreadsheet[15] is available here to help process the data generated by short-term baildown tests and obtain an LNAPL transmissivity estimate. Short-term baildown tests are the most commonly used method.
- Manual Skimming Method: LNAPL is repeatedly recovered over time (on the order of several days), gauging the well as it recovers, and then initiating another recovery event before the LNAPL level rebounds by more than 25% of the original drawdown. This is done until the LNAPL removal rate stabilizes, and then this rate is used to calculate transmissivity.
- Long-Term Recovery-Based Methods: Typically applied to sites with on-going LNAPL recovery. There are several equations which can be used to calculate LNAPL transmissivity from the long-term recovery data.
- Tracer-Based Method: An emerging, less common method involving placing fluorescent dyes that are soluble in LNAPL but not water into a well. The dye concentration is measured over time to calculate the LNAPL flux through the well.
Two comparative studies have shown that different LNAPL transmissivity measurement techniques yield similar results[16][13]). For sites with more complex conditions such as perched LNAPL, confined LNAPL, and changes in potentiometric surface, see ASTM Standard E2856-13[14] and Kirkman et al. (2013)[18].
In addition to field testing, LNAPL transmissivity can also be estimated using an appropriate computer-based model, although it is best to calibrate the model to observed field values initially. Please see LNAPL Mobility and Recovery Models (below) for more on this topic.
Why is LNAPL mobility important?
All LNAPL starts out mobile during the release and the resulting pore-invading process, often referred to as “drainage” conditions (because as the LNAPL enters a pore, the water or air is “drained” out). LNAPL must displace fluids such as water and air to invade the pores. While larger pores in a given soil can be easily entered by LNAPL that has relatively low driving head (pressure) relative to water or air, smaller pores require larger heads relative to those fluids. This behavior results in incomplete LNAPL saturation of the pores, as shown in Figure 1.
Concerns with LNAPL mobility are primarily focused on preventing additional migration and on reduction of the source volume. Consider a release of LNAPL to the subsurface. During the invasion process LNAPL is migrating down to the water table while spreading. Pores at the front of the migration are interconnected as saturation is increasing. Hence all LNAPL is mobile. Once the release stops, pores near the release point and in the center of the LNAPL body will have more LNAPL flowing out of them than is flowing in. LNAPL saturations decrease as pores are evacuated of LNAPL. During this evacuation (also referred to as “imbibition”) the pores become disconnected and some LNAPL is left in smaller, now disconnected, pores. This phenomenon is due to capillary forces inhibiting gravity drain/hydraulic gradient mechanisms. This is similar to the fraction of water that will not drain out of a sponge. This behavior, a key feature of multiphase flow in porous media, results in incomplete removal of LNAPL from the soil and the formation of residual LNAPL (which can be conceptualized as a disconnected single LNAPL blob in a single pore). The fraction of LNAPL retained is termed “residual LNAPL saturation” and is a function of the pore size distribution or the soil type, the maximum saturation LNAPL initially achieved and the water saturation.
Multiple researchers[20][21] have investigated the range of expected values of residual saturation in soils at LNAPL release sites. The overall conclusion of this work is that residual saturation depends on the amount of LNAPL initially present in the pores (initial saturation, Sni), the soil type and the water saturation. Figure 5 illustrates how residual saturation values in the vadose zone vary with soil type and initial LNAPL saturation[21]. Key points from Figure 5 include: a) finer-grained soils like silts (ML) and clays (CL) often have higher residual saturation values than sands (SC or SP); and b) higher initial saturation values result in higher residual saturation values.
LNAPL Mobility and Recovery Models
LNAPL distribution and mobility can be evaluated using public domain computer models. The LNAPL Distribution and Recovery Model estimates LNAPL transmissivity, saturation and overall volume in the subsurface and aids in assessing the potential effectiveness of recovery efforts to reduce LNAPL source volume. The Hydrocarbon Spill Screening Model is a US EPA model for evaluating the infiltration of LNAPL following a release and the resulting risks as LNAPL reaches the water-table and spreads laterally.
- API LNAPL Distribution and Recovery Model (LDRM), API 4760[2] – Addresses the recoverability of LNAPL in terms of time, induced drawdown, well spacing and residual fraction.
- Hydrocarbon Spill Screening Model (HSSM)[22] – Evaluates potential risks of releases and promotes continuing improvement towards prevention.
LNAPL Mobility and Risk
Residual LNAPL poses the same risk of groundwater contamination and petroleum vapor intrusion as mobile LNAPL[1]. The LNAPL composition, not the LNAPL thickness or saturation in soil, drives the concentrations in vapor and groundwater phases and therefore the risk to receptors. Once most of the LNAPL is immobile and residual LNAPL dominates the source area, removal of any small remaining mobile fraction is not expected to reduce risks of groundwater contamination or petroleum vapor intrusion in a meaningful way.
Summary and Key Conclusions
Mobile LNAPL can pose a risk where continued migration and expansion of the LNAPL footprint is occurring. LNAPL recovery efforts should focus on sites where LNAPL migration has the potential to expand the LNAPL footprint and where LNAPL recovery can significantly reduce the overall source volume. Where most of the LNAPL is immobile, recovery of a small amount of mobile LNAPL is not expected to substantially reduce risks of groundwater contamination or petroleum vapor intrusion.
The ITRC (2018)[1] discusses lines of evidence for evaluating LNAPL Migration risk as well as the benefits of LNAPL recovery in source reduction. Often LNAPL sites exhibiting low transmissivity values (i.e. 0.1 to 0.8 ft2/day) or with stable LNAPL bodies that are comprised of mostly residual LNAPL will not benefit from recovery attempts to remove mobile LNAPL.
References
- ^ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 1.9 ITRC, 2018. LNAPL Site Management: LCSM evolution, decision process, and remedial technologies (LNAPL-3). Interstate Technical and Regulatory Council https://lnapl-3.itrcweb.org/
- ^ 2.0 2.1 Charbeneau, R.J., 2007. LNAPL Distribution and Recovery Model. Distribution and Recovery of Petroleum Hydrocarbon Liquids in Porous Media. Vol. 1. API Publication 4760. Report.pdf https://www.api.org/oil-and-natural-gas/environment/clean-water/ground-water/lnapl/ldrm
- ^ 3.0 3.1 CRC CARE, 2018. Technical measurement guidance for LNAPL natural source zone depletion. Cooperative Research Centre for Contamination Assessment and Remediation of the Environment, Newcastle, Australia. Technical Report no. 44. 254p Report.pdf https://www.crccare.com/files/dmfile/CRCCARETechnicalreport44_TechnicalmeasurementguidanceforLNAPLnaturalsourcezonedepletion.pdf
- ^ Mahler, N., Sale, T. and Lyverse, M., 2012. A mass balance approach to resolving LNAPL stability. Groundwater, 50(6), pp.861-871. [10.1111/j.1745-6584.2012.00949.x 10.1111/j.1745-6584.2012.00949.x
- ^ Hawkins, A. M., 2013. Processes Controlling the behavior of LNAPLs at groundwater surface water interfaces, Master of Science Thesis, Colorado State University, Ft. Collins, Colorado. Report.pdf
- ^ 6.0 6.1 Huntley, D., 2000. Analytic determination of hydrocarbon transmissivity from baildown tests. Groundwater, 38(1), pp.46-52. doi: 10.1111/j.1745-6584.2000.tb00201.x
- ^ 7.0 7.1 USEPA, 1996. How to effectively recover free product at leaking underground storage sites. A guide for state regulators. USEPA 510-R-96-001. U.S. Environmental Protection Agency, 162 pp. Report.pdf
- ^ Newell, C. J., Acree*, S. D., Ross, R.R., and Huling, S.G. 1995. Light non-aqueous phase liquid. U.S. Environmental Protection Agency, Washington, DC. EPA/540/S-95/500 (NTIS 95-267738) Report.pdf
- ^ Williams, D.E. and Wilder, D.G., 1971. Gasoline Pollution of a Ground‐Water Reservoir—A Case History. Groundwater, 9(6), pp.50-56. doi: 10.1111/j.1745-6584.1971.tb03577.x
- ^ Mercer, J.W. and Cohen, R.M., 1990. A review of immiscible fluids in the subsurface: properties, models, characterization and remediation. Journal of contaminant hydrology, 6(2), pp.107-163. doi: 10.1016/0169-7722(90)90043-G
- ^ Kolhatkar, R., Kremesec, V., Rubin, S., Yukawa, C. and Senn, R., 1999. Application of field and analytical techniques to evaluate recoverability of subsurface free phase hydrocarbons. Petroleum Hydrocarbons and Organic Chemicals in Ground Water, pp.5-15.
- ^ Lundy, D.A. and Zimmerman, L.M., 1996, May. Assessing the recoverability of LNAPL plumes for recovery system conceptual design. In Proceedings of the 10th National Outdoor Action Conference and Expo (pp. 13-15).
- ^ 13.0 13.1 Kirkman, A.J., 2013. Refinement of Bouwer‐Rice baildown test analysis. Groundwater Monitoring & Remediation, 33(1), pp.105-110. doi: 10.1111/j.1745-6592.2012.01411.x
- ^ 14.0 14.1 14.2 ASTM, 2013. ASTM E2856-13, Standard guide for estimation of LNAPL transmissivity, ASTM International, West Conshohocken, PA.
- ^ 15.0 15.1 Charbeneau, R., Kirkman, A., and Muthu, R., 2016. API LNAPL Transmissivity Workbook: A Tool for Baildown Test Analysis – User Guide. American Petroleum Institute Publication 4762.
- ^ 16.0 16.1 Palmier, C., Dodt, M. and Atteia, O., 2016. Comparison of Oil Transmissivity Methods Using Bail‐Down Test Data. Groundwater Monitoring & Remediation, 36(3), pp.73-83. Report.pdf
- ^ Lenhard, R.J., Rayner, J.L. and Davis, G.B., 2017. A practical tool for estimating subsurface LNAPL distributions and transmissivity using current and historical fluid levels in groundwater wells: Effects of entrapped and residual LNAPL. Journal of contaminant hydrology, 205, pp.1-11. Report.pdf
- ^ 18.0 18.1 Kirkman, A.J., Adamski, M. and Hawthorne, J.M., 2013. Identification and assessment of confined and perched LNAPL conditions. Groundwater Monitoring & Remediation, 33(1), pp.75-86. doi: 10.1111/j.1745-6592.2012.01412.x
- ^ Gatsios, E., García-Rincón, J., Rayner, J.L., McLaughlan, R.G. and Davis, G.B., 2018. LNAPL transmissivity as a remediation metric in complex sites under water table fluctuations. Journal of Environmental Management, 215, pp.40-48. Report.pdf
- ^ Lenhard, R.J., Oostrom, M. and Dane, J.H., 2004. A constitutive model for air-NAPL-water flow in the vadose zone accounting for immobile, non-occluded (residual) NAPL in strongly water-wet porous media. Journal of Contaminant Hydrology, Vol. 73(1-4), pp 283-304 Report.pdf
- ^ 21.0 21.1 Johnston, C.D. and Adamski, M., 2005, August. Relationship between initial and residual LNAPL saturation for different soil types. In Proceedings of the 2005 Petroleum Hydrocarbons and Organic Chemicals in Groundwater®: Prevention, Assessment, and Remediation Conference (pp. 17-19). Report.pdf
- ^ Weaver, J.W., Charbeneau, R.J., Tauxe, J.D., Lien, B.K. and Provost, J.B., 1995. The Hydrocarbon spill screening model (HSSM) Volume 1: User’s Guide. US EPA, publication EPA/600/R-94/039a, 229pp. Report.pdf